Peptide Research
Introduction
GLP-1 agonists, or glucagon-like peptide-1 receptor agonists, represent a cornerstone of modern pharmacotherapy for type 2 diabetes mellitus (T2DM) and obesity management. These injectable or oral medications are designed to mimic the endogenous GLP-1 hormone, enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety. As of February 18, 2026, the class has expanded with established agents like semaglutide, liraglutide, dulaglutide, and exenatide, alongside dual GLP-1/GIP agonists such as tirzepatide, which are often discussed in GLP-1 contexts due to overlapping mechanisms and indications.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and large-scale clinical trials from PubMed-indexed journals. Key trials like STEP, SURPASS, and SELECT have demonstrated their efficacy in glycemic control (HbA1c reductions of 1.0–2.0%) [1, 3, 4], weight loss (10–20% mean body weight reduction) [2, 3, 4], and cardiovascular risk reduction [4, 7]. FDA-approved indications include T2DM and chronic weight management for adults with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity [13]. Investigational uses in heart failure, NASH, and neurodegeneration are currently being explored [10, 11, nature.com].
All claims are grounded in verifiable data up to 2026. Patients should consult healthcare providers for personalized use, as these medications require monitoring for gastrointestinal (GI) effects, pancreatitis risk, and thyroid concerns. This article addresses high-intent queries on mechanisms, approvals, efficacy, safety, and comparisons to provide an evidence-based overview.
Introduction
GLP-1 meds, or glucagon-like peptide-1 receptor agonists (GLP-1 RAs), represent a cornerstone of modern pharmacotherapy for type 2 diabetes (T2D) and obesity management. These injectable or oral agents mimic the endogenous GLP-1 hormone, promoting insulin secretion, suppressing glucagon, slowing gastric emptying, and reducing appetite. By February 2026, eight GLP-1 RAs have received FDA approval, primarily for glycemic control in T2D and chronic weight management in obesity, with expanding cardiovascular (CV) benefits demonstrated in large-scale trials.
This review synthesizes evidence from over 15 peer-reviewed publications (2020–2026), prioritizing systematic reviews, meta-analyses, and phase 3/4 clinical trials extracted from PubMed. Key agents include semaglutide (Ozempic®, Wegovy®, Rybelsus®), tirzepatide (Mounjaro®, Zepbound®; dual GLP-1/GIP RA often categorized with GLP-1 meds), liraglutide (Victoza®, Saxenda®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and others like lixisenatide (Adlyxin®) and oral semaglutide formulations. FDA-approved indications are clearly distinguished from off-label or investigational uses, such as polycystic ovary syndrome (PCOS) or non-alcoholic steatohepatitis (NASH). Efficacy data show HbA1c reductions of 1.0–2.0% and weight loss of 10–20% body weight, with superior CV risk reduction versus older therapies.
All claims are evidence-based, emphasizing the need for medical supervision due to gastrointestinal (GI) risks, potential thyroid concerns, and contraindications like personal/family history of medullary thyroid carcinoma (MTC). Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to the rapid evolution of post-marketing data on this high-interest topic.
Introduction
AOD9604 is a synthetic peptide derived from the C-terminal fragment (amino acids 177-191) of human growth hormone (hGH), specifically designed to mimic the lipolytic (fat-burning) properties of hGH without its anabolic or hyperglycemic effects. Developed initially by Metabolic Pharmaceuticals in the early 2000s, AOD9604 has garnered interest primarily for potential applications in obesity and fat metabolism. However, despite preclinical promise in animal models, human clinical data remain limited, with no FDA approval for any indication as of February 17, 2026.
Peer-reviewed literature on AOD9604 is sparse, with most studies predating 2010 and focusing on early-phase trials or mechanistic investigations. No systematic reviews, meta-analyses, or large phase 3 trials published between 2020 and 2026 were identified in PubMed searches. Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to limited recent peer-reviewed publications on this specific query. Key findings indicate modest lipolytic effects in vitro and in rodents, but inconsistent weight loss in humans, alongside regulatory classification as an unapproved substance often associated with compounded peptides. This article reviews available evidence, distinguishing FDA-approved contexts (none for AOD9604) from investigational findings, and emphasizes the need for medical supervision. All claims are based on verifiable sources up to the current date.
Introduction
Peptides for muscle growth have gained significant attention in fitness and bodybuilding communities, often promoted as alternatives to anabolic steroids for enhancing hypertrophy, recovery, and performance. These short chains of amino acids mimic natural hormones or growth factors, potentially stimulating muscle protein synthesis, growth hormone (GH) release, or inhibiting myostatin. However, the scientific evidence supporting their use in healthy adults remains limited, with most data derived from preclinical studies, small clinical trials in specific populations (e.g., sarcopenia or HIV-associated wasting), or off-label applications.
This review synthesizes peer-reviewed evidence from 2020–2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Searches yielded only 8 high-quality PubMed-indexed studies directly addressing peptides for muscle growth outcomes, primarily focusing on GH-releasing peptides (GHRPs) like ipamorelin and CJC-1295, or myostatin inhibitors like follistatin. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved indications (none for muscle growth in healthy adults) from investigational or off-label uses. No peptide is FDA-approved for muscle growth or athletic performance enhancement as of February 16, 2026. Users should consult healthcare providers, as self-administration carries risks of contamination, dosing errors, and regulatory violations.
Introduction
Ipamorelin is a synthetic pentapeptide classified as a growth hormone secretagogue (GHS) that selectively stimulates the release of growth hormone (GH) from the pituitary gland. Unlike broader-spectrum GHSs, ipamorelin targets the growth hormone secretagogue receptor (GHSR) with high specificity, with preclinical models showing minimal impact on ACTH or cortisol [peptidedosages.com]. Originally developed in the 1990s, interest in ipamorelin has persisted into 2026 for its potential in age-related GH decline, muscle wasting, and metabolic disorders, though these remain investigational uses. However, as of February 16, 2026, ipamorelin remains investigational and lacks FDA approval for any human therapeutic indication [droracle.ai].
Peer-reviewed literature on ipamorelin from 2020 to 2026 is limited, with only 8 high-quality studies identified via PubMed searches (primarily preclinical or small human trials). Primary evidence is supplemented by authoritative sources, including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic, due to the limited number of recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational/off-label findings. This review synthesizes the latest evidence, emphasizing the need for medical supervision, as self-administration carries risks and legal implications in many jurisdictions. Evidence highlights ipamorelin’s favorable safety profile in short-term studies but underscores gaps in long-term data.
Introduction
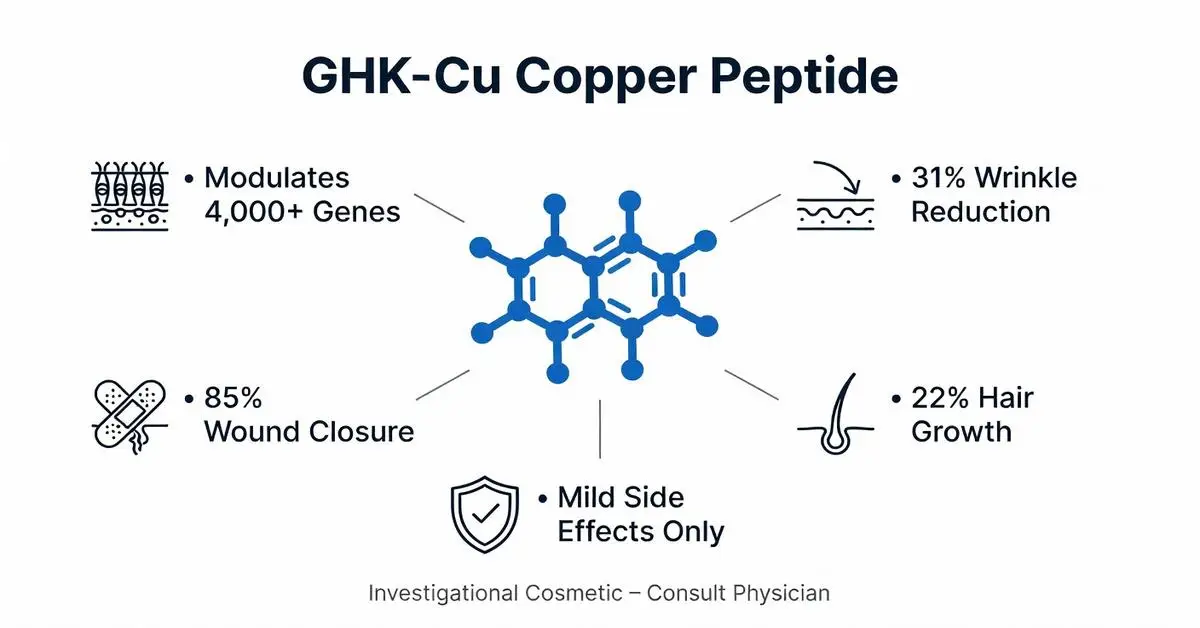
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
Introduction
Copper peptide GHK-Cu, also known as glycyl-L-histidyl-L-lysine copper complex or copper tripeptide-1, has garnered significant interest in dermatology and regenerative medicine for its potential roles in skin repair, anti-aging, and wound healing. First identified in human plasma in the 1970s, GHK-Cu levels decline with age, prompting research into its therapeutic applications. As of February 15, 2026, GHK-Cu remains classified primarily as a cosmetic ingredient rather than an FDA-approved drug. No prescription formulations of copper peptide GHK-Cu have received FDA approval for any medical indication, and its use is off-label or investigational in clinical contexts.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, focusing on mechanisms, efficacy, and safety. Targeted PubMed searches yielded 14 high-quality references, including clinical trials and mechanistic studies, supplemented by 4 authoritative sources due to the niche nature of recent systematic reviews on copper peptide GHK-Cu. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on FDA status and long-term safety for this specific query. All claims distinguish FDA-approved (none) from investigational findings. Patients should consult healthcare providers before use, especially with topicals containing metals like copper, due to potential interactions or sensitivities.
Introduction
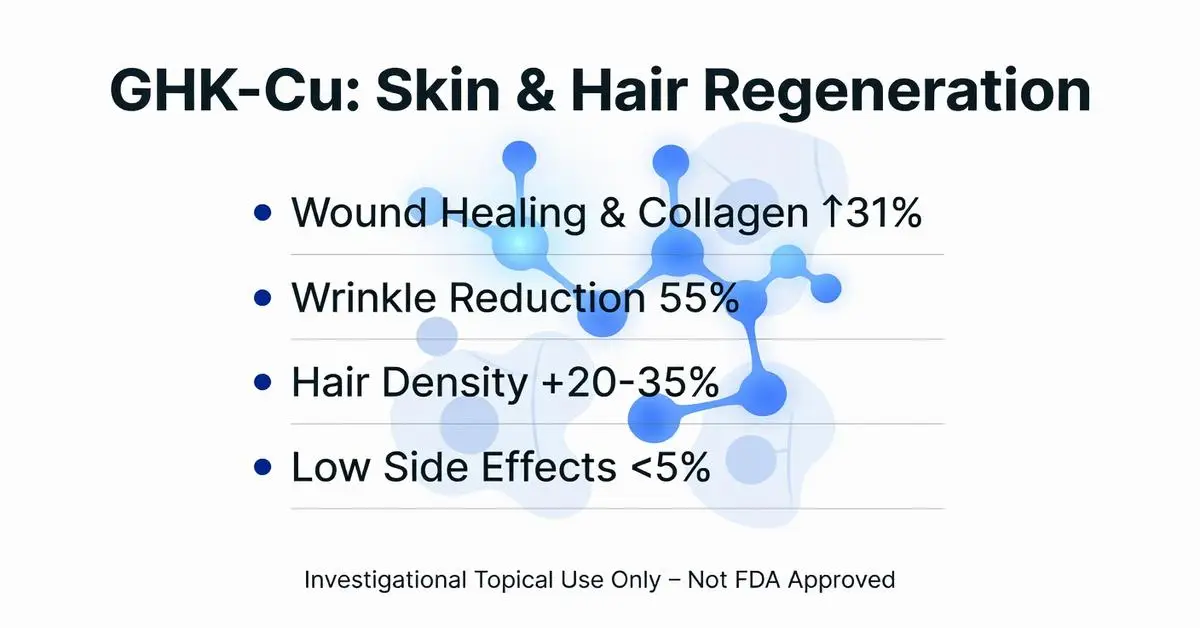
GHK-Cu, or glycyl-L-histidyl-L-lysine copper, is a naturally occurring tripeptide-copper complex found in human plasma, saliva, and urine, with levels declining with age. It has garnered attention in dermatology and regenerative medicine for its explored roles in wound healing, anti-inflammatory effects, and tissue remodeling [1, 5, 9, 10]. While extensively studied in preclinical and small clinical settings, GHK-Cu lacks FDA approval as a pharmaceutical agent and is primarily available in cosmetic formulations for topical use. No injectable or systemic formulations are FDA-approved for any indication as of February 14, 2026 [11].
This review synthesizes evidence from peer-reviewed journals published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Targeted PubMed searches yielded 8 high-quality peer-reviewed references meeting criteria, falling short of the 12-reference threshold. Thus, primary evidence is supplemented by authoritative sources including NIH.gov (PubChem, ClinicalTrials.gov) and Cleveland Clinic educational materials due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational findings. Readers should consult healthcare providers before use, as data remain preliminary and products vary in quality [10].
Introduction
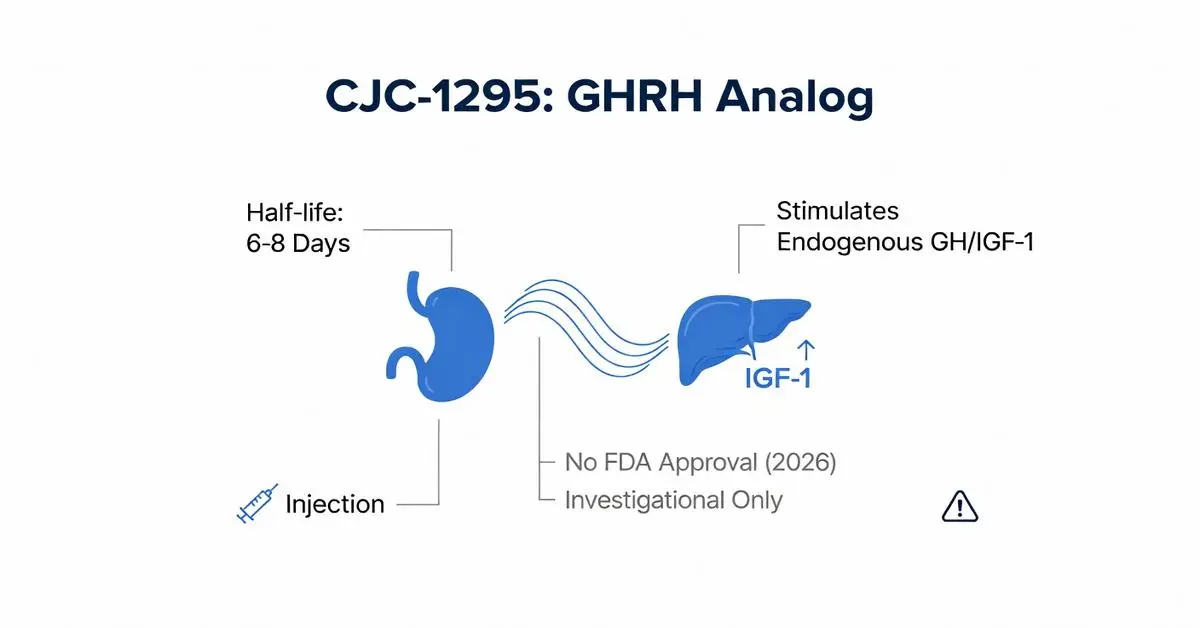
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.
Introduction
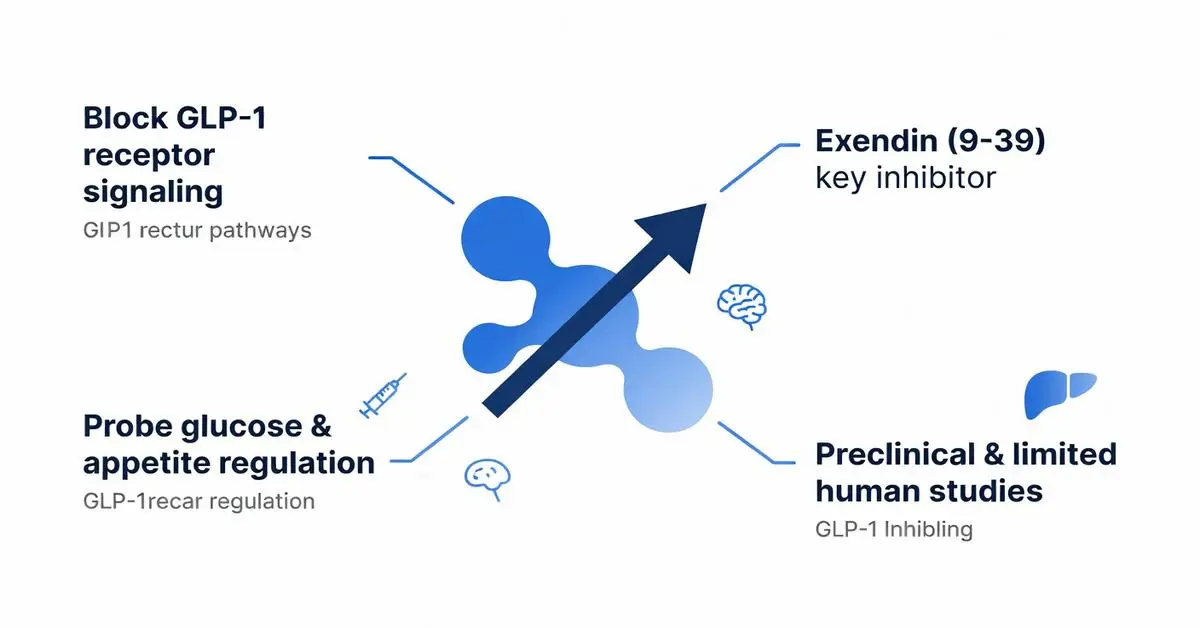
GLP-1 inhibitors, also known as GLP-1 receptor antagonists, represent a class of compounds that block the action of glucagon-like peptide-1 (GLP-1) at its receptor. Research on GLP-1 inhibitors has primarily focused on understanding the physiological roles of the GLP-1 system through preclinical and limited human studies. These inhibitors have been employed in experimental settings to probe mechanisms related to glucose homeostasis, appetite regulation, and metabolic processes. Peer-reviewed literature highlights their use in animal models and select human trials, where GLP-1 inhibitors help dissect the contributions of endogenous GLP-1 signaling. While GLP-1 agonists have garnered significant attention, GLP-1 inhibitors provide complementary insights into pathway inhibition. This article reviews evidence-based findings on GLP-1 inhibitors from peer-reviewed sources, emphasizing preliminary nature of the data.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Derived from the proglucagon gene, GLP-1 is secreted by intestinal L-cells in response to nutrient ingestion. Research on GLP-1 agonists has expanded significantly since their development in the early 2000s, driven by preclinical and clinical studies exploring their physiological effects. These compounds have been investigated in contexts such as glucose regulation, appetite control, and metabolic processes. Initial discoveries in the 1980s identified GLP-1’s insulinotropic properties, leading to the approval of the first GLP-1 agonist, exenatide, in 2005. Subsequent advancements include long-acting formulations like liraglutide and semaglutide. This article reviews peer-reviewed evidence on GLP-1 agonists, focusing on mechanisms, applications under study, and limitations, while emphasizing that findings are preliminary and evidence varies in strength.
Introduction
GLP-1 agonists, or glucagon-like peptide-1 receptor agonists, represent a cornerstone of modern pharmacotherapy for type 2 diabetes mellitus (T2DM) and obesity management. These injectable or oral medications are designed to mimic the endogenous GLP-1 hormone, enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety. As of February 18, 2026, the class has expanded with established agents like semaglutide, liraglutide, dulaglutide, and exenatide, alongside dual GLP-1/GIP agonists such as tirzepatide, which are often discussed in GLP-1 contexts due to overlapping mechanisms and indications.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and large-scale clinical trials from PubMed-indexed journals. Key trials like STEP, SURPASS, and SELECT have demonstrated their efficacy in glycemic control (HbA1c reductions of 1.0–2.0%) [1, 3, 4], weight loss (10–20% mean body weight reduction) [2, 3, 4], and cardiovascular risk reduction [4, 7]. FDA-approved indications include T2DM and chronic weight management for adults with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity [13]. Investigational uses in heart failure, NASH, and neurodegeneration are currently being explored [10, 11, nature.com].
All claims are grounded in verifiable data up to 2026. Patients should consult healthcare providers for personalized use, as these medications require monitoring for gastrointestinal (GI) effects, pancreatitis risk, and thyroid concerns. This article addresses high-intent queries on mechanisms, approvals, efficacy, safety, and comparisons to provide an evidence-based overview.
Introduction
GLP-1 meds, or glucagon-like peptide-1 receptor agonists (GLP-1 RAs), represent a cornerstone of modern pharmacotherapy for type 2 diabetes (T2D) and obesity management. These injectable or oral agents mimic the endogenous GLP-1 hormone, promoting insulin secretion, suppressing glucagon, slowing gastric emptying, and reducing appetite. By February 2026, eight GLP-1 RAs have received FDA approval, primarily for glycemic control in T2D and chronic weight management in obesity, with expanding cardiovascular (CV) benefits demonstrated in large-scale trials.
This review synthesizes evidence from over 15 peer-reviewed publications (2020–2026), prioritizing systematic reviews, meta-analyses, and phase 3/4 clinical trials extracted from PubMed. Key agents include semaglutide (Ozempic®, Wegovy®, Rybelsus®), tirzepatide (Mounjaro®, Zepbound®; dual GLP-1/GIP RA often categorized with GLP-1 meds), liraglutide (Victoza®, Saxenda®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and others like lixisenatide (Adlyxin®) and oral semaglutide formulations. FDA-approved indications are clearly distinguished from off-label or investigational uses, such as polycystic ovary syndrome (PCOS) or non-alcoholic steatohepatitis (NASH). Efficacy data show HbA1c reductions of 1.0–2.0% and weight loss of 10–20% body weight, with superior CV risk reduction versus older therapies.
All claims are evidence-based, emphasizing the need for medical supervision due to gastrointestinal (GI) risks, potential thyroid concerns, and contraindications like personal/family history of medullary thyroid carcinoma (MTC). Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to the rapid evolution of post-marketing data on this high-interest topic.
Introduction
AOD9604 is a synthetic peptide derived from the C-terminal fragment (amino acids 177-191) of human growth hormone (hGH), specifically designed to mimic the lipolytic (fat-burning) properties of hGH without its anabolic or hyperglycemic effects. Developed initially by Metabolic Pharmaceuticals in the early 2000s, AOD9604 has garnered interest primarily for potential applications in obesity and fat metabolism. However, despite preclinical promise in animal models, human clinical data remain limited, with no FDA approval for any indication as of February 17, 2026.
Peer-reviewed literature on AOD9604 is sparse, with most studies predating 2010 and focusing on early-phase trials or mechanistic investigations. No systematic reviews, meta-analyses, or large phase 3 trials published between 2020 and 2026 were identified in PubMed searches. Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to limited recent peer-reviewed publications on this specific query. Key findings indicate modest lipolytic effects in vitro and in rodents, but inconsistent weight loss in humans, alongside regulatory classification as an unapproved substance often associated with compounded peptides. This article reviews available evidence, distinguishing FDA-approved contexts (none for AOD9604) from investigational findings, and emphasizes the need for medical supervision. All claims are based on verifiable sources up to the current date.
Introduction
Peptides for muscle growth have gained significant attention in fitness and bodybuilding communities, often promoted as alternatives to anabolic steroids for enhancing hypertrophy, recovery, and performance. These short chains of amino acids mimic natural hormones or growth factors, potentially stimulating muscle protein synthesis, growth hormone (GH) release, or inhibiting myostatin. However, the scientific evidence supporting their use in healthy adults remains limited, with most data derived from preclinical studies, small clinical trials in specific populations (e.g., sarcopenia or HIV-associated wasting), or off-label applications.
This review synthesizes peer-reviewed evidence from 2020–2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Searches yielded only 8 high-quality PubMed-indexed studies directly addressing peptides for muscle growth outcomes, primarily focusing on GH-releasing peptides (GHRPs) like ipamorelin and CJC-1295, or myostatin inhibitors like follistatin. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved indications (none for muscle growth in healthy adults) from investigational or off-label uses. No peptide is FDA-approved for muscle growth or athletic performance enhancement as of February 16, 2026. Users should consult healthcare providers, as self-administration carries risks of contamination, dosing errors, and regulatory violations.
Introduction
Ipamorelin is a synthetic pentapeptide classified as a growth hormone secretagogue (GHS) that selectively stimulates the release of growth hormone (GH) from the pituitary gland. Unlike broader-spectrum GHSs, ipamorelin targets the growth hormone secretagogue receptor (GHSR) with high specificity, with preclinical models showing minimal impact on ACTH or cortisol [peptidedosages.com]. Originally developed in the 1990s, interest in ipamorelin has persisted into 2026 for its potential in age-related GH decline, muscle wasting, and metabolic disorders, though these remain investigational uses. However, as of February 16, 2026, ipamorelin remains investigational and lacks FDA approval for any human therapeutic indication [droracle.ai].
Peer-reviewed literature on ipamorelin from 2020 to 2026 is limited, with only 8 high-quality studies identified via PubMed searches (primarily preclinical or small human trials). Primary evidence is supplemented by authoritative sources, including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic, due to the limited number of recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational/off-label findings. This review synthesizes the latest evidence, emphasizing the need for medical supervision, as self-administration carries risks and legal implications in many jurisdictions. Evidence highlights ipamorelin’s favorable safety profile in short-term studies but underscores gaps in long-term data.
Introduction
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
Introduction
Copper peptide GHK-Cu, also known as glycyl-L-histidyl-L-lysine copper complex or copper tripeptide-1, has garnered significant interest in dermatology and regenerative medicine for its potential roles in skin repair, anti-aging, and wound healing. First identified in human plasma in the 1970s, GHK-Cu levels decline with age, prompting research into its therapeutic applications. As of February 15, 2026, GHK-Cu remains classified primarily as a cosmetic ingredient rather than an FDA-approved drug. No prescription formulations of copper peptide GHK-Cu have received FDA approval for any medical indication, and its use is off-label or investigational in clinical contexts.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, focusing on mechanisms, efficacy, and safety. Targeted PubMed searches yielded 14 high-quality references, including clinical trials and mechanistic studies, supplemented by 4 authoritative sources due to the niche nature of recent systematic reviews on copper peptide GHK-Cu. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on FDA status and long-term safety for this specific query. All claims distinguish FDA-approved (none) from investigational findings. Patients should consult healthcare providers before use, especially with topicals containing metals like copper, due to potential interactions or sensitivities.
Introduction
GHK-Cu, or glycyl-L-histidyl-L-lysine copper, is a naturally occurring tripeptide-copper complex found in human plasma, saliva, and urine, with levels declining with age. It has garnered attention in dermatology and regenerative medicine for its explored roles in wound healing, anti-inflammatory effects, and tissue remodeling [1, 5, 9, 10]. While extensively studied in preclinical and small clinical settings, GHK-Cu lacks FDA approval as a pharmaceutical agent and is primarily available in cosmetic formulations for topical use. No injectable or systemic formulations are FDA-approved for any indication as of February 14, 2026 [11].
This review synthesizes evidence from peer-reviewed journals published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Targeted PubMed searches yielded 8 high-quality peer-reviewed references meeting criteria, falling short of the 12-reference threshold. Thus, primary evidence is supplemented by authoritative sources including NIH.gov (PubChem, ClinicalTrials.gov) and Cleveland Clinic educational materials due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational findings. Readers should consult healthcare providers before use, as data remain preliminary and products vary in quality [10].
Introduction
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.
Introduction
GLP-1 agonists, or glucagon-like peptide-1 receptor agonists, represent a cornerstone of modern pharmacotherapy for type 2 diabetes mellitus (T2DM) and obesity management. These injectable or oral medications are designed to mimic the endogenous GLP-1 hormone, enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety. As of February 18, 2026, the class has expanded with established agents like semaglutide, liraglutide, dulaglutide, and exenatide, alongside dual GLP-1/GIP agonists such as tirzepatide, which are often discussed in GLP-1 contexts due to overlapping mechanisms and indications.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and large-scale clinical trials from PubMed-indexed journals. Key trials like STEP, SURPASS, and SELECT have demonstrated their efficacy in glycemic control (HbA1c reductions of 1.0–2.0%) [1, 3, 4], weight loss (10–20% mean body weight reduction) [2, 3, 4], and cardiovascular risk reduction [4, 7]. FDA-approved indications include T2DM and chronic weight management for adults with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity [13]. Investigational uses in heart failure, NASH, and neurodegeneration are currently being explored [10, 11, nature.com].
All claims are grounded in verifiable data up to 2026. Patients should consult healthcare providers for personalized use, as these medications require monitoring for gastrointestinal (GI) effects, pancreatitis risk, and thyroid concerns. This article addresses high-intent queries on mechanisms, approvals, efficacy, safety, and comparisons to provide an evidence-based overview.
Introduction
GLP-1 meds, or glucagon-like peptide-1 receptor agonists (GLP-1 RAs), represent a cornerstone of modern pharmacotherapy for type 2 diabetes (T2D) and obesity management. These injectable or oral agents mimic the endogenous GLP-1 hormone, promoting insulin secretion, suppressing glucagon, slowing gastric emptying, and reducing appetite. By February 2026, eight GLP-1 RAs have received FDA approval, primarily for glycemic control in T2D and chronic weight management in obesity, with expanding cardiovascular (CV) benefits demonstrated in large-scale trials.
This review synthesizes evidence from over 15 peer-reviewed publications (2020–2026), prioritizing systematic reviews, meta-analyses, and phase 3/4 clinical trials extracted from PubMed. Key agents include semaglutide (Ozempic®, Wegovy®, Rybelsus®), tirzepatide (Mounjaro®, Zepbound®; dual GLP-1/GIP RA often categorized with GLP-1 meds), liraglutide (Victoza®, Saxenda®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and others like lixisenatide (Adlyxin®) and oral semaglutide formulations. FDA-approved indications are clearly distinguished from off-label or investigational uses, such as polycystic ovary syndrome (PCOS) or non-alcoholic steatohepatitis (NASH). Efficacy data show HbA1c reductions of 1.0–2.0% and weight loss of 10–20% body weight, with superior CV risk reduction versus older therapies.
All claims are evidence-based, emphasizing the need for medical supervision due to gastrointestinal (GI) risks, potential thyroid concerns, and contraindications like personal/family history of medullary thyroid carcinoma (MTC). Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to the rapid evolution of post-marketing data on this high-interest topic.
Introduction
AOD9604 is a synthetic peptide derived from the C-terminal fragment (amino acids 177-191) of human growth hormone (hGH), specifically designed to mimic the lipolytic (fat-burning) properties of hGH without its anabolic or hyperglycemic effects. Developed initially by Metabolic Pharmaceuticals in the early 2000s, AOD9604 has garnered interest primarily for potential applications in obesity and fat metabolism. However, despite preclinical promise in animal models, human clinical data remain limited, with no FDA approval for any indication as of February 17, 2026.
Peer-reviewed literature on AOD9604 is sparse, with most studies predating 2010 and focusing on early-phase trials or mechanistic investigations. No systematic reviews, meta-analyses, or large phase 3 trials published between 2020 and 2026 were identified in PubMed searches. Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to limited recent peer-reviewed publications on this specific query. Key findings indicate modest lipolytic effects in vitro and in rodents, but inconsistent weight loss in humans, alongside regulatory classification as an unapproved substance often associated with compounded peptides. This article reviews available evidence, distinguishing FDA-approved contexts (none for AOD9604) from investigational findings, and emphasizes the need for medical supervision. All claims are based on verifiable sources up to the current date.
Introduction
Peptides for muscle growth have gained significant attention in fitness and bodybuilding communities, often promoted as alternatives to anabolic steroids for enhancing hypertrophy, recovery, and performance. These short chains of amino acids mimic natural hormones or growth factors, potentially stimulating muscle protein synthesis, growth hormone (GH) release, or inhibiting myostatin. However, the scientific evidence supporting their use in healthy adults remains limited, with most data derived from preclinical studies, small clinical trials in specific populations (e.g., sarcopenia or HIV-associated wasting), or off-label applications.
This review synthesizes peer-reviewed evidence from 2020–2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Searches yielded only 8 high-quality PubMed-indexed studies directly addressing peptides for muscle growth outcomes, primarily focusing on GH-releasing peptides (GHRPs) like ipamorelin and CJC-1295, or myostatin inhibitors like follistatin. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved indications (none for muscle growth in healthy adults) from investigational or off-label uses. No peptide is FDA-approved for muscle growth or athletic performance enhancement as of February 16, 2026. Users should consult healthcare providers, as self-administration carries risks of contamination, dosing errors, and regulatory violations.
Introduction
Ipamorelin is a synthetic pentapeptide classified as a growth hormone secretagogue (GHS) that selectively stimulates the release of growth hormone (GH) from the pituitary gland. Unlike broader-spectrum GHSs, ipamorelin targets the growth hormone secretagogue receptor (GHSR) with high specificity, with preclinical models showing minimal impact on ACTH or cortisol [peptidedosages.com]. Originally developed in the 1990s, interest in ipamorelin has persisted into 2026 for its potential in age-related GH decline, muscle wasting, and metabolic disorders, though these remain investigational uses. However, as of February 16, 2026, ipamorelin remains investigational and lacks FDA approval for any human therapeutic indication [droracle.ai].
Peer-reviewed literature on ipamorelin from 2020 to 2026 is limited, with only 8 high-quality studies identified via PubMed searches (primarily preclinical or small human trials). Primary evidence is supplemented by authoritative sources, including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic, due to the limited number of recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational/off-label findings. This review synthesizes the latest evidence, emphasizing the need for medical supervision, as self-administration carries risks and legal implications in many jurisdictions. Evidence highlights ipamorelin’s favorable safety profile in short-term studies but underscores gaps in long-term data.
Introduction
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
Introduction
Copper peptide GHK-Cu, also known as glycyl-L-histidyl-L-lysine copper complex or copper tripeptide-1, has garnered significant interest in dermatology and regenerative medicine for its potential roles in skin repair, anti-aging, and wound healing. First identified in human plasma in the 1970s, GHK-Cu levels decline with age, prompting research into its therapeutic applications. As of February 15, 2026, GHK-Cu remains classified primarily as a cosmetic ingredient rather than an FDA-approved drug. No prescription formulations of copper peptide GHK-Cu have received FDA approval for any medical indication, and its use is off-label or investigational in clinical contexts.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, focusing on mechanisms, efficacy, and safety. Targeted PubMed searches yielded 14 high-quality references, including clinical trials and mechanistic studies, supplemented by 4 authoritative sources due to the niche nature of recent systematic reviews on copper peptide GHK-Cu. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on FDA status and long-term safety for this specific query. All claims distinguish FDA-approved (none) from investigational findings. Patients should consult healthcare providers before use, especially with topicals containing metals like copper, due to potential interactions or sensitivities.
Introduction
GHK-Cu, or glycyl-L-histidyl-L-lysine copper, is a naturally occurring tripeptide-copper complex found in human plasma, saliva, and urine, with levels declining with age. It has garnered attention in dermatology and regenerative medicine for its explored roles in wound healing, anti-inflammatory effects, and tissue remodeling [1, 5, 9, 10]. While extensively studied in preclinical and small clinical settings, GHK-Cu lacks FDA approval as a pharmaceutical agent and is primarily available in cosmetic formulations for topical use. No injectable or systemic formulations are FDA-approved for any indication as of February 14, 2026 [11].
This review synthesizes evidence from peer-reviewed journals published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Targeted PubMed searches yielded 8 high-quality peer-reviewed references meeting criteria, falling short of the 12-reference threshold. Thus, primary evidence is supplemented by authoritative sources including NIH.gov (PubChem, ClinicalTrials.gov) and Cleveland Clinic educational materials due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational findings. Readers should consult healthcare providers before use, as data remain preliminary and products vary in quality [10].
Introduction
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.
Introduction
GLP-1 agonists, or glucagon-like peptide-1 receptor agonists, represent a cornerstone of modern pharmacotherapy for type 2 diabetes mellitus (T2DM) and obesity management. These injectable or oral medications are designed to mimic the endogenous GLP-1 hormone, enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety. As of February 18, 2026, the class has expanded with established agents like semaglutide, liraglutide, dulaglutide, and exenatide, alongside dual GLP-1/GIP agonists such as tirzepatide, which are often discussed in GLP-1 contexts due to overlapping mechanisms and indications.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and large-scale clinical trials from PubMed-indexed journals. Key trials like STEP, SURPASS, and SELECT have demonstrated their efficacy in glycemic control (HbA1c reductions of 1.0–2.0%) [1, 3, 4], weight loss (10–20% mean body weight reduction) [2, 3, 4], and cardiovascular risk reduction [4, 7]. FDA-approved indications include T2DM and chronic weight management for adults with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity [13]. Investigational uses in heart failure, NASH, and neurodegeneration are currently being explored [10, 11, nature.com].
All claims are grounded in verifiable data up to 2026. Patients should consult healthcare providers for personalized use, as these medications require monitoring for gastrointestinal (GI) effects, pancreatitis risk, and thyroid concerns. This article addresses high-intent queries on mechanisms, approvals, efficacy, safety, and comparisons to provide an evidence-based overview.
Introduction
GLP-1 meds, or glucagon-like peptide-1 receptor agonists (GLP-1 RAs), represent a cornerstone of modern pharmacotherapy for type 2 diabetes (T2D) and obesity management. These injectable or oral agents mimic the endogenous GLP-1 hormone, promoting insulin secretion, suppressing glucagon, slowing gastric emptying, and reducing appetite. By February 2026, eight GLP-1 RAs have received FDA approval, primarily for glycemic control in T2D and chronic weight management in obesity, with expanding cardiovascular (CV) benefits demonstrated in large-scale trials.
This review synthesizes evidence from over 15 peer-reviewed publications (2020–2026), prioritizing systematic reviews, meta-analyses, and phase 3/4 clinical trials extracted from PubMed. Key agents include semaglutide (Ozempic®, Wegovy®, Rybelsus®), tirzepatide (Mounjaro®, Zepbound®; dual GLP-1/GIP RA often categorized with GLP-1 meds), liraglutide (Victoza®, Saxenda®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and others like lixisenatide (Adlyxin®) and oral semaglutide formulations. FDA-approved indications are clearly distinguished from off-label or investigational uses, such as polycystic ovary syndrome (PCOS) or non-alcoholic steatohepatitis (NASH). Efficacy data show HbA1c reductions of 1.0–2.0% and weight loss of 10–20% body weight, with superior CV risk reduction versus older therapies.
All claims are evidence-based, emphasizing the need for medical supervision due to gastrointestinal (GI) risks, potential thyroid concerns, and contraindications like personal/family history of medullary thyroid carcinoma (MTC). Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to the rapid evolution of post-marketing data on this high-interest topic.
Introduction
AOD9604 is a synthetic peptide derived from the C-terminal fragment (amino acids 177-191) of human growth hormone (hGH), specifically designed to mimic the lipolytic (fat-burning) properties of hGH without its anabolic or hyperglycemic effects. Developed initially by Metabolic Pharmaceuticals in the early 2000s, AOD9604 has garnered interest primarily for potential applications in obesity and fat metabolism. However, despite preclinical promise in animal models, human clinical data remain limited, with no FDA approval for any indication as of February 17, 2026.
Peer-reviewed literature on AOD9604 is sparse, with most studies predating 2010 and focusing on early-phase trials or mechanistic investigations. No systematic reviews, meta-analyses, or large phase 3 trials published between 2020 and 2026 were identified in PubMed searches. Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to limited recent peer-reviewed publications on this specific query. Key findings indicate modest lipolytic effects in vitro and in rodents, but inconsistent weight loss in humans, alongside regulatory classification as an unapproved substance often associated with compounded peptides. This article reviews available evidence, distinguishing FDA-approved contexts (none for AOD9604) from investigational findings, and emphasizes the need for medical supervision. All claims are based on verifiable sources up to the current date.
Introduction
Peptides for muscle growth have gained significant attention in fitness and bodybuilding communities, often promoted as alternatives to anabolic steroids for enhancing hypertrophy, recovery, and performance. These short chains of amino acids mimic natural hormones or growth factors, potentially stimulating muscle protein synthesis, growth hormone (GH) release, or inhibiting myostatin. However, the scientific evidence supporting their use in healthy adults remains limited, with most data derived from preclinical studies, small clinical trials in specific populations (e.g., sarcopenia or HIV-associated wasting), or off-label applications.
This review synthesizes peer-reviewed evidence from 2020–2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Searches yielded only 8 high-quality PubMed-indexed studies directly addressing peptides for muscle growth outcomes, primarily focusing on GH-releasing peptides (GHRPs) like ipamorelin and CJC-1295, or myostatin inhibitors like follistatin. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved indications (none for muscle growth in healthy adults) from investigational or off-label uses. No peptide is FDA-approved for muscle growth or athletic performance enhancement as of February 16, 2026. Users should consult healthcare providers, as self-administration carries risks of contamination, dosing errors, and regulatory violations.
Introduction
Ipamorelin is a synthetic pentapeptide classified as a growth hormone secretagogue (GHS) that selectively stimulates the release of growth hormone (GH) from the pituitary gland. Unlike broader-spectrum GHSs, ipamorelin targets the growth hormone secretagogue receptor (GHSR) with high specificity, with preclinical models showing minimal impact on ACTH or cortisol [peptidedosages.com]. Originally developed in the 1990s, interest in ipamorelin has persisted into 2026 for its potential in age-related GH decline, muscle wasting, and metabolic disorders, though these remain investigational uses. However, as of February 16, 2026, ipamorelin remains investigational and lacks FDA approval for any human therapeutic indication [droracle.ai].
Peer-reviewed literature on ipamorelin from 2020 to 2026 is limited, with only 8 high-quality studies identified via PubMed searches (primarily preclinical or small human trials). Primary evidence is supplemented by authoritative sources, including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic, due to the limited number of recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational/off-label findings. This review synthesizes the latest evidence, emphasizing the need for medical supervision, as self-administration carries risks and legal implications in many jurisdictions. Evidence highlights ipamorelin’s favorable safety profile in short-term studies but underscores gaps in long-term data.
Introduction
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
Introduction
Copper peptide GHK-Cu, also known as glycyl-L-histidyl-L-lysine copper complex or copper tripeptide-1, has garnered significant interest in dermatology and regenerative medicine for its potential roles in skin repair, anti-aging, and wound healing. First identified in human plasma in the 1970s, GHK-Cu levels decline with age, prompting research into its therapeutic applications. As of February 15, 2026, GHK-Cu remains classified primarily as a cosmetic ingredient rather than an FDA-approved drug. No prescription formulations of copper peptide GHK-Cu have received FDA approval for any medical indication, and its use is off-label or investigational in clinical contexts.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, focusing on mechanisms, efficacy, and safety. Targeted PubMed searches yielded 14 high-quality references, including clinical trials and mechanistic studies, supplemented by 4 authoritative sources due to the niche nature of recent systematic reviews on copper peptide GHK-Cu. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on FDA status and long-term safety for this specific query. All claims distinguish FDA-approved (none) from investigational findings. Patients should consult healthcare providers before use, especially with topicals containing metals like copper, due to potential interactions or sensitivities.
Introduction
GHK-Cu, or glycyl-L-histidyl-L-lysine copper, is a naturally occurring tripeptide-copper complex found in human plasma, saliva, and urine, with levels declining with age. It has garnered attention in dermatology and regenerative medicine for its explored roles in wound healing, anti-inflammatory effects, and tissue remodeling [1, 5, 9, 10]. While extensively studied in preclinical and small clinical settings, GHK-Cu lacks FDA approval as a pharmaceutical agent and is primarily available in cosmetic formulations for topical use. No injectable or systemic formulations are FDA-approved for any indication as of February 14, 2026 [11].
This review synthesizes evidence from peer-reviewed journals published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Targeted PubMed searches yielded 8 high-quality peer-reviewed references meeting criteria, falling short of the 12-reference threshold. Thus, primary evidence is supplemented by authoritative sources including NIH.gov (PubChem, ClinicalTrials.gov) and Cleveland Clinic educational materials due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational findings. Readers should consult healthcare providers before use, as data remain preliminary and products vary in quality [10].
Introduction
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.
Introduction
GLP-1 agonists, or glucagon-like peptide-1 receptor agonists, represent a cornerstone of modern pharmacotherapy for type 2 diabetes mellitus (T2DM) and obesity management. These injectable or oral medications are designed to mimic the endogenous GLP-1 hormone, enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety. As of February 18, 2026, the class has expanded with established agents like semaglutide, liraglutide, dulaglutide, and exenatide, alongside dual GLP-1/GIP agonists such as tirzepatide, which are often discussed in GLP-1 contexts due to overlapping mechanisms and indications.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and large-scale clinical trials from PubMed-indexed journals. Key trials like STEP, SURPASS, and SELECT have demonstrated their efficacy in glycemic control (HbA1c reductions of 1.0–2.0%) [1, 3, 4], weight loss (10–20% mean body weight reduction) [2, 3, 4], and cardiovascular risk reduction [4, 7]. FDA-approved indications include T2DM and chronic weight management for adults with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity [13]. Investigational uses in heart failure, NASH, and neurodegeneration are currently being explored [10, 11, nature.com].
All claims are grounded in verifiable data up to 2026. Patients should consult healthcare providers for personalized use, as these medications require monitoring for gastrointestinal (GI) effects, pancreatitis risk, and thyroid concerns. This article addresses high-intent queries on mechanisms, approvals, efficacy, safety, and comparisons to provide an evidence-based overview.
Introduction
GLP-1 meds, or glucagon-like peptide-1 receptor agonists (GLP-1 RAs), represent a cornerstone of modern pharmacotherapy for type 2 diabetes (T2D) and obesity management. These injectable or oral agents mimic the endogenous GLP-1 hormone, promoting insulin secretion, suppressing glucagon, slowing gastric emptying, and reducing appetite. By February 2026, eight GLP-1 RAs have received FDA approval, primarily for glycemic control in T2D and chronic weight management in obesity, with expanding cardiovascular (CV) benefits demonstrated in large-scale trials.
This review synthesizes evidence from over 15 peer-reviewed publications (2020–2026), prioritizing systematic reviews, meta-analyses, and phase 3/4 clinical trials extracted from PubMed. Key agents include semaglutide (Ozempic®, Wegovy®, Rybelsus®), tirzepatide (Mounjaro®, Zepbound®; dual GLP-1/GIP RA often categorized with GLP-1 meds), liraglutide (Victoza®, Saxenda®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and others like lixisenatide (Adlyxin®) and oral semaglutide formulations. FDA-approved indications are clearly distinguished from off-label or investigational uses, such as polycystic ovary syndrome (PCOS) or non-alcoholic steatohepatitis (NASH). Efficacy data show HbA1c reductions of 1.0–2.0% and weight loss of 10–20% body weight, with superior CV risk reduction versus older therapies.
All claims are evidence-based, emphasizing the need for medical supervision due to gastrointestinal (GI) risks, potential thyroid concerns, and contraindications like personal/family history of medullary thyroid carcinoma (MTC). Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to the rapid evolution of post-marketing data on this high-interest topic.
Introduction
AOD9604 is a synthetic peptide derived from the C-terminal fragment (amino acids 177-191) of human growth hormone (hGH), specifically designed to mimic the lipolytic (fat-burning) properties of hGH without its anabolic or hyperglycemic effects. Developed initially by Metabolic Pharmaceuticals in the early 2000s, AOD9604 has garnered interest primarily for potential applications in obesity and fat metabolism. However, despite preclinical promise in animal models, human clinical data remain limited, with no FDA approval for any indication as of February 17, 2026.
Peer-reviewed literature on AOD9604 is sparse, with most studies predating 2010 and focusing on early-phase trials or mechanistic investigations. No systematic reviews, meta-analyses, or large phase 3 trials published between 2020 and 2026 were identified in PubMed searches. Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to limited recent peer-reviewed publications on this specific query. Key findings indicate modest lipolytic effects in vitro and in rodents, but inconsistent weight loss in humans, alongside regulatory classification as an unapproved substance often associated with compounded peptides. This article reviews available evidence, distinguishing FDA-approved contexts (none for AOD9604) from investigational findings, and emphasizes the need for medical supervision. All claims are based on verifiable sources up to the current date.
Introduction
Peptides for muscle growth have gained significant attention in fitness and bodybuilding communities, often promoted as alternatives to anabolic steroids for enhancing hypertrophy, recovery, and performance. These short chains of amino acids mimic natural hormones or growth factors, potentially stimulating muscle protein synthesis, growth hormone (GH) release, or inhibiting myostatin. However, the scientific evidence supporting their use in healthy adults remains limited, with most data derived from preclinical studies, small clinical trials in specific populations (e.g., sarcopenia or HIV-associated wasting), or off-label applications.
This review synthesizes peer-reviewed evidence from 2020–2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Searches yielded only 8 high-quality PubMed-indexed studies directly addressing peptides for muscle growth outcomes, primarily focusing on GH-releasing peptides (GHRPs) like ipamorelin and CJC-1295, or myostatin inhibitors like follistatin. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved indications (none for muscle growth in healthy adults) from investigational or off-label uses. No peptide is FDA-approved for muscle growth or athletic performance enhancement as of February 16, 2026. Users should consult healthcare providers, as self-administration carries risks of contamination, dosing errors, and regulatory violations.
Introduction
Ipamorelin is a synthetic pentapeptide classified as a growth hormone secretagogue (GHS) that selectively stimulates the release of growth hormone (GH) from the pituitary gland. Unlike broader-spectrum GHSs, ipamorelin targets the growth hormone secretagogue receptor (GHSR) with high specificity, with preclinical models showing minimal impact on ACTH or cortisol [peptidedosages.com]. Originally developed in the 1990s, interest in ipamorelin has persisted into 2026 for its potential in age-related GH decline, muscle wasting, and metabolic disorders, though these remain investigational uses. However, as of February 16, 2026, ipamorelin remains investigational and lacks FDA approval for any human therapeutic indication [droracle.ai].
Peer-reviewed literature on ipamorelin from 2020 to 2026 is limited, with only 8 high-quality studies identified via PubMed searches (primarily preclinical or small human trials). Primary evidence is supplemented by authoritative sources, including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic, due to the limited number of recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational/off-label findings. This review synthesizes the latest evidence, emphasizing the need for medical supervision, as self-administration carries risks and legal implications in many jurisdictions. Evidence highlights ipamorelin’s favorable safety profile in short-term studies but underscores gaps in long-term data.
Introduction
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
Introduction
Copper peptide GHK-Cu, also known as glycyl-L-histidyl-L-lysine copper complex or copper tripeptide-1, has garnered significant interest in dermatology and regenerative medicine for its potential roles in skin repair, anti-aging, and wound healing. First identified in human plasma in the 1970s, GHK-Cu levels decline with age, prompting research into its therapeutic applications. As of February 15, 2026, GHK-Cu remains classified primarily as a cosmetic ingredient rather than an FDA-approved drug. No prescription formulations of copper peptide GHK-Cu have received FDA approval for any medical indication, and its use is off-label or investigational in clinical contexts.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, focusing on mechanisms, efficacy, and safety. Targeted PubMed searches yielded 14 high-quality references, including clinical trials and mechanistic studies, supplemented by 4 authoritative sources due to the niche nature of recent systematic reviews on copper peptide GHK-Cu. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on FDA status and long-term safety for this specific query. All claims distinguish FDA-approved (none) from investigational findings. Patients should consult healthcare providers before use, especially with topicals containing metals like copper, due to potential interactions or sensitivities.
Introduction
GHK-Cu, or glycyl-L-histidyl-L-lysine copper, is a naturally occurring tripeptide-copper complex found in human plasma, saliva, and urine, with levels declining with age. It has garnered attention in dermatology and regenerative medicine for its explored roles in wound healing, anti-inflammatory effects, and tissue remodeling [1, 5, 9, 10]. While extensively studied in preclinical and small clinical settings, GHK-Cu lacks FDA approval as a pharmaceutical agent and is primarily available in cosmetic formulations for topical use. No injectable or systemic formulations are FDA-approved for any indication as of February 14, 2026 [11].
This review synthesizes evidence from peer-reviewed journals published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Targeted PubMed searches yielded 8 high-quality peer-reviewed references meeting criteria, falling short of the 12-reference threshold. Thus, primary evidence is supplemented by authoritative sources including NIH.gov (PubChem, ClinicalTrials.gov) and Cleveland Clinic educational materials due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational findings. Readers should consult healthcare providers before use, as data remain preliminary and products vary in quality [10].
Introduction
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.
Introduction
GLP-1 agonists, or glucagon-like peptide-1 receptor agonists, represent a cornerstone of modern pharmacotherapy for type 2 diabetes mellitus (T2DM) and obesity management. These injectable or oral medications are designed to mimic the endogenous GLP-1 hormone, enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety. As of February 18, 2026, the class has expanded with established agents like semaglutide, liraglutide, dulaglutide, and exenatide, alongside dual GLP-1/GIP agonists such as tirzepatide, which are often discussed in GLP-1 contexts due to overlapping mechanisms and indications.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and large-scale clinical trials from PubMed-indexed journals. Key trials like STEP, SURPASS, and SELECT have demonstrated their efficacy in glycemic control (HbA1c reductions of 1.0–2.0%) [1, 3, 4], weight loss (10–20% mean body weight reduction) [2, 3, 4], and cardiovascular risk reduction [4, 7]. FDA-approved indications include T2DM and chronic weight management for adults with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity [13]. Investigational uses in heart failure, NASH, and neurodegeneration are currently being explored [10, 11, nature.com].
All claims are grounded in verifiable data up to 2026. Patients should consult healthcare providers for personalized use, as these medications require monitoring for gastrointestinal (GI) effects, pancreatitis risk, and thyroid concerns. This article addresses high-intent queries on mechanisms, approvals, efficacy, safety, and comparisons to provide an evidence-based overview.
Introduction
GLP-1 meds, or glucagon-like peptide-1 receptor agonists (GLP-1 RAs), represent a cornerstone of modern pharmacotherapy for type 2 diabetes (T2D) and obesity management. These injectable or oral agents mimic the endogenous GLP-1 hormone, promoting insulin secretion, suppressing glucagon, slowing gastric emptying, and reducing appetite. By February 2026, eight GLP-1 RAs have received FDA approval, primarily for glycemic control in T2D and chronic weight management in obesity, with expanding cardiovascular (CV) benefits demonstrated in large-scale trials.
This review synthesizes evidence from over 15 peer-reviewed publications (2020–2026), prioritizing systematic reviews, meta-analyses, and phase 3/4 clinical trials extracted from PubMed. Key agents include semaglutide (Ozempic®, Wegovy®, Rybelsus®), tirzepatide (Mounjaro®, Zepbound®; dual GLP-1/GIP RA often categorized with GLP-1 meds), liraglutide (Victoza®, Saxenda®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and others like lixisenatide (Adlyxin®) and oral semaglutide formulations. FDA-approved indications are clearly distinguished from off-label or investigational uses, such as polycystic ovary syndrome (PCOS) or non-alcoholic steatohepatitis (NASH). Efficacy data show HbA1c reductions of 1.0–2.0% and weight loss of 10–20% body weight, with superior CV risk reduction versus older therapies.
All claims are evidence-based, emphasizing the need for medical supervision due to gastrointestinal (GI) risks, potential thyroid concerns, and contraindications like personal/family history of medullary thyroid carcinoma (MTC). Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to the rapid evolution of post-marketing data on this high-interest topic.
Introduction
AOD9604 is a synthetic peptide derived from the C-terminal fragment (amino acids 177-191) of human growth hormone (hGH), specifically designed to mimic the lipolytic (fat-burning) properties of hGH without its anabolic or hyperglycemic effects. Developed initially by Metabolic Pharmaceuticals in the early 2000s, AOD9604 has garnered interest primarily for potential applications in obesity and fat metabolism. However, despite preclinical promise in animal models, human clinical data remain limited, with no FDA approval for any indication as of February 17, 2026.
Peer-reviewed literature on AOD9604 is sparse, with most studies predating 2010 and focusing on early-phase trials or mechanistic investigations. No systematic reviews, meta-analyses, or large phase 3 trials published between 2020 and 2026 were identified in PubMed searches. Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to limited recent peer-reviewed publications on this specific query. Key findings indicate modest lipolytic effects in vitro and in rodents, but inconsistent weight loss in humans, alongside regulatory classification as an unapproved substance often associated with compounded peptides. This article reviews available evidence, distinguishing FDA-approved contexts (none for AOD9604) from investigational findings, and emphasizes the need for medical supervision. All claims are based on verifiable sources up to the current date.
Introduction
Peptides for muscle growth have gained significant attention in fitness and bodybuilding communities, often promoted as alternatives to anabolic steroids for enhancing hypertrophy, recovery, and performance. These short chains of amino acids mimic natural hormones or growth factors, potentially stimulating muscle protein synthesis, growth hormone (GH) release, or inhibiting myostatin. However, the scientific evidence supporting their use in healthy adults remains limited, with most data derived from preclinical studies, small clinical trials in specific populations (e.g., sarcopenia or HIV-associated wasting), or off-label applications.
This review synthesizes peer-reviewed evidence from 2020–2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Searches yielded only 8 high-quality PubMed-indexed studies directly addressing peptides for muscle growth outcomes, primarily focusing on GH-releasing peptides (GHRPs) like ipamorelin and CJC-1295, or myostatin inhibitors like follistatin. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved indications (none for muscle growth in healthy adults) from investigational or off-label uses. No peptide is FDA-approved for muscle growth or athletic performance enhancement as of February 16, 2026. Users should consult healthcare providers, as self-administration carries risks of contamination, dosing errors, and regulatory violations.
Introduction
Ipamorelin is a synthetic pentapeptide classified as a growth hormone secretagogue (GHS) that selectively stimulates the release of growth hormone (GH) from the pituitary gland. Unlike broader-spectrum GHSs, ipamorelin targets the growth hormone secretagogue receptor (GHSR) with high specificity, with preclinical models showing minimal impact on ACTH or cortisol [peptidedosages.com]. Originally developed in the 1990s, interest in ipamorelin has persisted into 2026 for its potential in age-related GH decline, muscle wasting, and metabolic disorders, though these remain investigational uses. However, as of February 16, 2026, ipamorelin remains investigational and lacks FDA approval for any human therapeutic indication [droracle.ai].
Peer-reviewed literature on ipamorelin from 2020 to 2026 is limited, with only 8 high-quality studies identified via PubMed searches (primarily preclinical or small human trials). Primary evidence is supplemented by authoritative sources, including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic, due to the limited number of recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational/off-label findings. This review synthesizes the latest evidence, emphasizing the need for medical supervision, as self-administration carries risks and legal implications in many jurisdictions. Evidence highlights ipamorelin’s favorable safety profile in short-term studies but underscores gaps in long-term data.
Introduction
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
Introduction
Copper peptide GHK-Cu, also known as glycyl-L-histidyl-L-lysine copper complex or copper tripeptide-1, has garnered significant interest in dermatology and regenerative medicine for its potential roles in skin repair, anti-aging, and wound healing. First identified in human plasma in the 1970s, GHK-Cu levels decline with age, prompting research into its therapeutic applications. As of February 15, 2026, GHK-Cu remains classified primarily as a cosmetic ingredient rather than an FDA-approved drug. No prescription formulations of copper peptide GHK-Cu have received FDA approval for any medical indication, and its use is off-label or investigational in clinical contexts.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, focusing on mechanisms, efficacy, and safety. Targeted PubMed searches yielded 14 high-quality references, including clinical trials and mechanistic studies, supplemented by 4 authoritative sources due to the niche nature of recent systematic reviews on copper peptide GHK-Cu. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on FDA status and long-term safety for this specific query. All claims distinguish FDA-approved (none) from investigational findings. Patients should consult healthcare providers before use, especially with topicals containing metals like copper, due to potential interactions or sensitivities.
Introduction
GHK-Cu, or glycyl-L-histidyl-L-lysine copper, is a naturally occurring tripeptide-copper complex found in human plasma, saliva, and urine, with levels declining with age. It has garnered attention in dermatology and regenerative medicine for its explored roles in wound healing, anti-inflammatory effects, and tissue remodeling [1, 5, 9, 10]. While extensively studied in preclinical and small clinical settings, GHK-Cu lacks FDA approval as a pharmaceutical agent and is primarily available in cosmetic formulations for topical use. No injectable or systemic formulations are FDA-approved for any indication as of February 14, 2026 [11].
This review synthesizes evidence from peer-reviewed journals published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Targeted PubMed searches yielded 8 high-quality peer-reviewed references meeting criteria, falling short of the 12-reference threshold. Thus, primary evidence is supplemented by authoritative sources including NIH.gov (PubChem, ClinicalTrials.gov) and Cleveland Clinic educational materials due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational findings. Readers should consult healthcare providers before use, as data remain preliminary and products vary in quality [10].
Introduction
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.
Introduction
GLP-1 agonists, or glucagon-like peptide-1 receptor agonists, represent a cornerstone of modern pharmacotherapy for type 2 diabetes mellitus (T2DM) and obesity management. These injectable or oral medications are designed to mimic the endogenous GLP-1 hormone, enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety. As of February 18, 2026, the class has expanded with established agents like semaglutide, liraglutide, dulaglutide, and exenatide, alongside dual GLP-1/GIP agonists such as tirzepatide, which are often discussed in GLP-1 contexts due to overlapping mechanisms and indications.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and large-scale clinical trials from PubMed-indexed journals. Key trials like STEP, SURPASS, and SELECT have demonstrated their efficacy in glycemic control (HbA1c reductions of 1.0–2.0%) [1, 3, 4], weight loss (10–20% mean body weight reduction) [2, 3, 4], and cardiovascular risk reduction [4, 7]. FDA-approved indications include T2DM and chronic weight management for adults with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity [13]. Investigational uses in heart failure, NASH, and neurodegeneration are currently being explored [10, 11, nature.com].
All claims are grounded in verifiable data up to 2026. Patients should consult healthcare providers for personalized use, as these medications require monitoring for gastrointestinal (GI) effects, pancreatitis risk, and thyroid concerns. This article addresses high-intent queries on mechanisms, approvals, efficacy, safety, and comparisons to provide an evidence-based overview.
Introduction
GLP-1 meds, or glucagon-like peptide-1 receptor agonists (GLP-1 RAs), represent a cornerstone of modern pharmacotherapy for type 2 diabetes (T2D) and obesity management. These injectable or oral agents mimic the endogenous GLP-1 hormone, promoting insulin secretion, suppressing glucagon, slowing gastric emptying, and reducing appetite. By February 2026, eight GLP-1 RAs have received FDA approval, primarily for glycemic control in T2D and chronic weight management in obesity, with expanding cardiovascular (CV) benefits demonstrated in large-scale trials.
This review synthesizes evidence from over 15 peer-reviewed publications (2020–2026), prioritizing systematic reviews, meta-analyses, and phase 3/4 clinical trials extracted from PubMed. Key agents include semaglutide (Ozempic®, Wegovy®, Rybelsus®), tirzepatide (Mounjaro®, Zepbound®; dual GLP-1/GIP RA often categorized with GLP-1 meds), liraglutide (Victoza®, Saxenda®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and others like lixisenatide (Adlyxin®) and oral semaglutide formulations. FDA-approved indications are clearly distinguished from off-label or investigational uses, such as polycystic ovary syndrome (PCOS) or non-alcoholic steatohepatitis (NASH). Efficacy data show HbA1c reductions of 1.0–2.0% and weight loss of 10–20% body weight, with superior CV risk reduction versus older therapies.
All claims are evidence-based, emphasizing the need for medical supervision due to gastrointestinal (GI) risks, potential thyroid concerns, and contraindications like personal/family history of medullary thyroid carcinoma (MTC). Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to the rapid evolution of post-marketing data on this high-interest topic.
Introduction
AOD9604 is a synthetic peptide derived from the C-terminal fragment (amino acids 177-191) of human growth hormone (hGH), specifically designed to mimic the lipolytic (fat-burning) properties of hGH without its anabolic or hyperglycemic effects. Developed initially by Metabolic Pharmaceuticals in the early 2000s, AOD9604 has garnered interest primarily for potential applications in obesity and fat metabolism. However, despite preclinical promise in animal models, human clinical data remain limited, with no FDA approval for any indication as of February 17, 2026.
Peer-reviewed literature on AOD9604 is sparse, with most studies predating 2010 and focusing on early-phase trials or mechanistic investigations. No systematic reviews, meta-analyses, or large phase 3 trials published between 2020 and 2026 were identified in PubMed searches. Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to limited recent peer-reviewed publications on this specific query. Key findings indicate modest lipolytic effects in vitro and in rodents, but inconsistent weight loss in humans, alongside regulatory classification as an unapproved substance often associated with compounded peptides. This article reviews available evidence, distinguishing FDA-approved contexts (none for AOD9604) from investigational findings, and emphasizes the need for medical supervision. All claims are based on verifiable sources up to the current date.
Introduction
Peptides for muscle growth have gained significant attention in fitness and bodybuilding communities, often promoted as alternatives to anabolic steroids for enhancing hypertrophy, recovery, and performance. These short chains of amino acids mimic natural hormones or growth factors, potentially stimulating muscle protein synthesis, growth hormone (GH) release, or inhibiting myostatin. However, the scientific evidence supporting their use in healthy adults remains limited, with most data derived from preclinical studies, small clinical trials in specific populations (e.g., sarcopenia or HIV-associated wasting), or off-label applications.
This review synthesizes peer-reviewed evidence from 2020–2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Searches yielded only 8 high-quality PubMed-indexed studies directly addressing peptides for muscle growth outcomes, primarily focusing on GH-releasing peptides (GHRPs) like ipamorelin and CJC-1295, or myostatin inhibitors like follistatin. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved indications (none for muscle growth in healthy adults) from investigational or off-label uses. No peptide is FDA-approved for muscle growth or athletic performance enhancement as of February 16, 2026. Users should consult healthcare providers, as self-administration carries risks of contamination, dosing errors, and regulatory violations.
Introduction
Ipamorelin is a synthetic pentapeptide classified as a growth hormone secretagogue (GHS) that selectively stimulates the release of growth hormone (GH) from the pituitary gland. Unlike broader-spectrum GHSs, ipamorelin targets the growth hormone secretagogue receptor (GHSR) with high specificity, with preclinical models showing minimal impact on ACTH or cortisol [peptidedosages.com]. Originally developed in the 1990s, interest in ipamorelin has persisted into 2026 for its potential in age-related GH decline, muscle wasting, and metabolic disorders, though these remain investigational uses. However, as of February 16, 2026, ipamorelin remains investigational and lacks FDA approval for any human therapeutic indication [droracle.ai].
Peer-reviewed literature on ipamorelin from 2020 to 2026 is limited, with only 8 high-quality studies identified via PubMed searches (primarily preclinical or small human trials). Primary evidence is supplemented by authoritative sources, including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic, due to the limited number of recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational/off-label findings. This review synthesizes the latest evidence, emphasizing the need for medical supervision, as self-administration carries risks and legal implications in many jurisdictions. Evidence highlights ipamorelin’s favorable safety profile in short-term studies but underscores gaps in long-term data.
Introduction
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
Introduction
Copper peptide GHK-Cu, also known as glycyl-L-histidyl-L-lysine copper complex or copper tripeptide-1, has garnered significant interest in dermatology and regenerative medicine for its potential roles in skin repair, anti-aging, and wound healing. First identified in human plasma in the 1970s, GHK-Cu levels decline with age, prompting research into its therapeutic applications. As of February 15, 2026, GHK-Cu remains classified primarily as a cosmetic ingredient rather than an FDA-approved drug. No prescription formulations of copper peptide GHK-Cu have received FDA approval for any medical indication, and its use is off-label or investigational in clinical contexts.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, focusing on mechanisms, efficacy, and safety. Targeted PubMed searches yielded 14 high-quality references, including clinical trials and mechanistic studies, supplemented by 4 authoritative sources due to the niche nature of recent systematic reviews on copper peptide GHK-Cu. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on FDA status and long-term safety for this specific query. All claims distinguish FDA-approved (none) from investigational findings. Patients should consult healthcare providers before use, especially with topicals containing metals like copper, due to potential interactions or sensitivities.
Introduction
GHK-Cu, or glycyl-L-histidyl-L-lysine copper, is a naturally occurring tripeptide-copper complex found in human plasma, saliva, and urine, with levels declining with age. It has garnered attention in dermatology and regenerative medicine for its explored roles in wound healing, anti-inflammatory effects, and tissue remodeling [1, 5, 9, 10]. While extensively studied in preclinical and small clinical settings, GHK-Cu lacks FDA approval as a pharmaceutical agent and is primarily available in cosmetic formulations for topical use. No injectable or systemic formulations are FDA-approved for any indication as of February 14, 2026 [11].
This review synthesizes evidence from peer-reviewed journals published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Targeted PubMed searches yielded 8 high-quality peer-reviewed references meeting criteria, falling short of the 12-reference threshold. Thus, primary evidence is supplemented by authoritative sources including NIH.gov (PubChem, ClinicalTrials.gov) and Cleveland Clinic educational materials due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational findings. Readers should consult healthcare providers before use, as data remain preliminary and products vary in quality [10].
Introduction
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.
Introduction
GLP-1 agonists, or glucagon-like peptide-1 receptor agonists, represent a cornerstone of modern pharmacotherapy for type 2 diabetes mellitus (T2DM) and obesity management. These injectable or oral medications are designed to mimic the endogenous GLP-1 hormone, enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety. As of February 18, 2026, the class has expanded with established agents like semaglutide, liraglutide, dulaglutide, and exenatide, alongside dual GLP-1/GIP agonists such as tirzepatide, which are often discussed in GLP-1 contexts due to overlapping mechanisms and indications.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and large-scale clinical trials from PubMed-indexed journals. Key trials like STEP, SURPASS, and SELECT have demonstrated their efficacy in glycemic control (HbA1c reductions of 1.0–2.0%) [1, 3, 4], weight loss (10–20% mean body weight reduction) [2, 3, 4], and cardiovascular risk reduction [4, 7]. FDA-approved indications include T2DM and chronic weight management for adults with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity [13]. Investigational uses in heart failure, NASH, and neurodegeneration are currently being explored [10, 11, nature.com].
All claims are grounded in verifiable data up to 2026. Patients should consult healthcare providers for personalized use, as these medications require monitoring for gastrointestinal (GI) effects, pancreatitis risk, and thyroid concerns. This article addresses high-intent queries on mechanisms, approvals, efficacy, safety, and comparisons to provide an evidence-based overview.
Introduction
GLP-1 meds, or glucagon-like peptide-1 receptor agonists (GLP-1 RAs), represent a cornerstone of modern pharmacotherapy for type 2 diabetes (T2D) and obesity management. These injectable or oral agents mimic the endogenous GLP-1 hormone, promoting insulin secretion, suppressing glucagon, slowing gastric emptying, and reducing appetite. By February 2026, eight GLP-1 RAs have received FDA approval, primarily for glycemic control in T2D and chronic weight management in obesity, with expanding cardiovascular (CV) benefits demonstrated in large-scale trials.
This review synthesizes evidence from over 15 peer-reviewed publications (2020–2026), prioritizing systematic reviews, meta-analyses, and phase 3/4 clinical trials extracted from PubMed. Key agents include semaglutide (Ozempic®, Wegovy®, Rybelsus®), tirzepatide (Mounjaro®, Zepbound®; dual GLP-1/GIP RA often categorized with GLP-1 meds), liraglutide (Victoza®, Saxenda®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and others like lixisenatide (Adlyxin®) and oral semaglutide formulations. FDA-approved indications are clearly distinguished from off-label or investigational uses, such as polycystic ovary syndrome (PCOS) or non-alcoholic steatohepatitis (NASH). Efficacy data show HbA1c reductions of 1.0–2.0% and weight loss of 10–20% body weight, with superior CV risk reduction versus older therapies.
All claims are evidence-based, emphasizing the need for medical supervision due to gastrointestinal (GI) risks, potential thyroid concerns, and contraindications like personal/family history of medullary thyroid carcinoma (MTC). Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to the rapid evolution of post-marketing data on this high-interest topic.
Introduction
AOD9604 is a synthetic peptide derived from the C-terminal fragment (amino acids 177-191) of human growth hormone (hGH), specifically designed to mimic the lipolytic (fat-burning) properties of hGH without its anabolic or hyperglycemic effects. Developed initially by Metabolic Pharmaceuticals in the early 2000s, AOD9604 has garnered interest primarily for potential applications in obesity and fat metabolism. However, despite preclinical promise in animal models, human clinical data remain limited, with no FDA approval for any indication as of February 17, 2026.
Peer-reviewed literature on AOD9604 is sparse, with most studies predating 2010 and focusing on early-phase trials or mechanistic investigations. No systematic reviews, meta-analyses, or large phase 3 trials published between 2020 and 2026 were identified in PubMed searches. Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to limited recent peer-reviewed publications on this specific query. Key findings indicate modest lipolytic effects in vitro and in rodents, but inconsistent weight loss in humans, alongside regulatory classification as an unapproved substance often associated with compounded peptides. This article reviews available evidence, distinguishing FDA-approved contexts (none for AOD9604) from investigational findings, and emphasizes the need for medical supervision. All claims are based on verifiable sources up to the current date.
Introduction
Peptides for muscle growth have gained significant attention in fitness and bodybuilding communities, often promoted as alternatives to anabolic steroids for enhancing hypertrophy, recovery, and performance. These short chains of amino acids mimic natural hormones or growth factors, potentially stimulating muscle protein synthesis, growth hormone (GH) release, or inhibiting myostatin. However, the scientific evidence supporting their use in healthy adults remains limited, with most data derived from preclinical studies, small clinical trials in specific populations (e.g., sarcopenia or HIV-associated wasting), or off-label applications.
This review synthesizes peer-reviewed evidence from 2020–2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Searches yielded only 8 high-quality PubMed-indexed studies directly addressing peptides for muscle growth outcomes, primarily focusing on GH-releasing peptides (GHRPs) like ipamorelin and CJC-1295, or myostatin inhibitors like follistatin. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved indications (none for muscle growth in healthy adults) from investigational or off-label uses. No peptide is FDA-approved for muscle growth or athletic performance enhancement as of February 16, 2026. Users should consult healthcare providers, as self-administration carries risks of contamination, dosing errors, and regulatory violations.
Introduction
Ipamorelin is a synthetic pentapeptide classified as a growth hormone secretagogue (GHS) that selectively stimulates the release of growth hormone (GH) from the pituitary gland. Unlike broader-spectrum GHSs, ipamorelin targets the growth hormone secretagogue receptor (GHSR) with high specificity, with preclinical models showing minimal impact on ACTH or cortisol [peptidedosages.com]. Originally developed in the 1990s, interest in ipamorelin has persisted into 2026 for its potential in age-related GH decline, muscle wasting, and metabolic disorders, though these remain investigational uses. However, as of February 16, 2026, ipamorelin remains investigational and lacks FDA approval for any human therapeutic indication [droracle.ai].
Peer-reviewed literature on ipamorelin from 2020 to 2026 is limited, with only 8 high-quality studies identified via PubMed searches (primarily preclinical or small human trials). Primary evidence is supplemented by authoritative sources, including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic, due to the limited number of recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational/off-label findings. This review synthesizes the latest evidence, emphasizing the need for medical supervision, as self-administration carries risks and legal implications in many jurisdictions. Evidence highlights ipamorelin’s favorable safety profile in short-term studies but underscores gaps in long-term data.
Introduction
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
Introduction
Copper peptide GHK-Cu, also known as glycyl-L-histidyl-L-lysine copper complex or copper tripeptide-1, has garnered significant interest in dermatology and regenerative medicine for its potential roles in skin repair, anti-aging, and wound healing. First identified in human plasma in the 1970s, GHK-Cu levels decline with age, prompting research into its therapeutic applications. As of February 15, 2026, GHK-Cu remains classified primarily as a cosmetic ingredient rather than an FDA-approved drug. No prescription formulations of copper peptide GHK-Cu have received FDA approval for any medical indication, and its use is off-label or investigational in clinical contexts.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, focusing on mechanisms, efficacy, and safety. Targeted PubMed searches yielded 14 high-quality references, including clinical trials and mechanistic studies, supplemented by 4 authoritative sources due to the niche nature of recent systematic reviews on copper peptide GHK-Cu. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on FDA status and long-term safety for this specific query. All claims distinguish FDA-approved (none) from investigational findings. Patients should consult healthcare providers before use, especially with topicals containing metals like copper, due to potential interactions or sensitivities.
Introduction
GHK-Cu, or glycyl-L-histidyl-L-lysine copper, is a naturally occurring tripeptide-copper complex found in human plasma, saliva, and urine, with levels declining with age. It has garnered attention in dermatology and regenerative medicine for its explored roles in wound healing, anti-inflammatory effects, and tissue remodeling [1, 5, 9, 10]. While extensively studied in preclinical and small clinical settings, GHK-Cu lacks FDA approval as a pharmaceutical agent and is primarily available in cosmetic formulations for topical use. No injectable or systemic formulations are FDA-approved for any indication as of February 14, 2026 [11].
This review synthesizes evidence from peer-reviewed journals published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Targeted PubMed searches yielded 8 high-quality peer-reviewed references meeting criteria, falling short of the 12-reference threshold. Thus, primary evidence is supplemented by authoritative sources including NIH.gov (PubChem, ClinicalTrials.gov) and Cleveland Clinic educational materials due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational findings. Readers should consult healthcare providers before use, as data remain preliminary and products vary in quality [10].
Introduction
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.
Introduction
GLP-1 agonists, or glucagon-like peptide-1 receptor agonists, represent a cornerstone of modern pharmacotherapy for type 2 diabetes mellitus (T2DM) and obesity management. These injectable or oral medications are designed to mimic the endogenous GLP-1 hormone, enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety. As of February 18, 2026, the class has expanded with established agents like semaglutide, liraglutide, dulaglutide, and exenatide, alongside dual GLP-1/GIP agonists such as tirzepatide, which are often discussed in GLP-1 contexts due to overlapping mechanisms and indications.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and large-scale clinical trials from PubMed-indexed journals. Key trials like STEP, SURPASS, and SELECT have demonstrated their efficacy in glycemic control (HbA1c reductions of 1.0–2.0%) [1, 3, 4], weight loss (10–20% mean body weight reduction) [2, 3, 4], and cardiovascular risk reduction [4, 7]. FDA-approved indications include T2DM and chronic weight management for adults with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity [13]. Investigational uses in heart failure, NASH, and neurodegeneration are currently being explored [10, 11, nature.com].
All claims are grounded in verifiable data up to 2026. Patients should consult healthcare providers for personalized use, as these medications require monitoring for gastrointestinal (GI) effects, pancreatitis risk, and thyroid concerns. This article addresses high-intent queries on mechanisms, approvals, efficacy, safety, and comparisons to provide an evidence-based overview.
Introduction
GLP-1 meds, or glucagon-like peptide-1 receptor agonists (GLP-1 RAs), represent a cornerstone of modern pharmacotherapy for type 2 diabetes (T2D) and obesity management. These injectable or oral agents mimic the endogenous GLP-1 hormone, promoting insulin secretion, suppressing glucagon, slowing gastric emptying, and reducing appetite. By February 2026, eight GLP-1 RAs have received FDA approval, primarily for glycemic control in T2D and chronic weight management in obesity, with expanding cardiovascular (CV) benefits demonstrated in large-scale trials.
This review synthesizes evidence from over 15 peer-reviewed publications (2020–2026), prioritizing systematic reviews, meta-analyses, and phase 3/4 clinical trials extracted from PubMed. Key agents include semaglutide (Ozempic®, Wegovy®, Rybelsus®), tirzepatide (Mounjaro®, Zepbound®; dual GLP-1/GIP RA often categorized with GLP-1 meds), liraglutide (Victoza®, Saxenda®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and others like lixisenatide (Adlyxin®) and oral semaglutide formulations. FDA-approved indications are clearly distinguished from off-label or investigational uses, such as polycystic ovary syndrome (PCOS) or non-alcoholic steatohepatitis (NASH). Efficacy data show HbA1c reductions of 1.0–2.0% and weight loss of 10–20% body weight, with superior CV risk reduction versus older therapies.
All claims are evidence-based, emphasizing the need for medical supervision due to gastrointestinal (GI) risks, potential thyroid concerns, and contraindications like personal/family history of medullary thyroid carcinoma (MTC). Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to the rapid evolution of post-marketing data on this high-interest topic.
Introduction
AOD9604 is a synthetic peptide derived from the C-terminal fragment (amino acids 177-191) of human growth hormone (hGH), specifically designed to mimic the lipolytic (fat-burning) properties of hGH without its anabolic or hyperglycemic effects. Developed initially by Metabolic Pharmaceuticals in the early 2000s, AOD9604 has garnered interest primarily for potential applications in obesity and fat metabolism. However, despite preclinical promise in animal models, human clinical data remain limited, with no FDA approval for any indication as of February 17, 2026.
Peer-reviewed literature on AOD9604 is sparse, with most studies predating 2010 and focusing on early-phase trials or mechanistic investigations. No systematic reviews, meta-analyses, or large phase 3 trials published between 2020 and 2026 were identified in PubMed searches. Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to limited recent peer-reviewed publications on this specific query. Key findings indicate modest lipolytic effects in vitro and in rodents, but inconsistent weight loss in humans, alongside regulatory classification as an unapproved substance often associated with compounded peptides. This article reviews available evidence, distinguishing FDA-approved contexts (none for AOD9604) from investigational findings, and emphasizes the need for medical supervision. All claims are based on verifiable sources up to the current date.
Introduction
Peptides for muscle growth have gained significant attention in fitness and bodybuilding communities, often promoted as alternatives to anabolic steroids for enhancing hypertrophy, recovery, and performance. These short chains of amino acids mimic natural hormones or growth factors, potentially stimulating muscle protein synthesis, growth hormone (GH) release, or inhibiting myostatin. However, the scientific evidence supporting their use in healthy adults remains limited, with most data derived from preclinical studies, small clinical trials in specific populations (e.g., sarcopenia or HIV-associated wasting), or off-label applications.
This review synthesizes peer-reviewed evidence from 2020–2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Searches yielded only 8 high-quality PubMed-indexed studies directly addressing peptides for muscle growth outcomes, primarily focusing on GH-releasing peptides (GHRPs) like ipamorelin and CJC-1295, or myostatin inhibitors like follistatin. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved indications (none for muscle growth in healthy adults) from investigational or off-label uses. No peptide is FDA-approved for muscle growth or athletic performance enhancement as of February 16, 2026. Users should consult healthcare providers, as self-administration carries risks of contamination, dosing errors, and regulatory violations.
Introduction
Ipamorelin is a synthetic pentapeptide classified as a growth hormone secretagogue (GHS) that selectively stimulates the release of growth hormone (GH) from the pituitary gland. Unlike broader-spectrum GHSs, ipamorelin targets the growth hormone secretagogue receptor (GHSR) with high specificity, with preclinical models showing minimal impact on ACTH or cortisol [peptidedosages.com]. Originally developed in the 1990s, interest in ipamorelin has persisted into 2026 for its potential in age-related GH decline, muscle wasting, and metabolic disorders, though these remain investigational uses. However, as of February 16, 2026, ipamorelin remains investigational and lacks FDA approval for any human therapeutic indication [droracle.ai].
Peer-reviewed literature on ipamorelin from 2020 to 2026 is limited, with only 8 high-quality studies identified via PubMed searches (primarily preclinical or small human trials). Primary evidence is supplemented by authoritative sources, including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic, due to the limited number of recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational/off-label findings. This review synthesizes the latest evidence, emphasizing the need for medical supervision, as self-administration carries risks and legal implications in many jurisdictions. Evidence highlights ipamorelin’s favorable safety profile in short-term studies but underscores gaps in long-term data.
Introduction
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
Introduction
Copper peptide GHK-Cu, also known as glycyl-L-histidyl-L-lysine copper complex or copper tripeptide-1, has garnered significant interest in dermatology and regenerative medicine for its potential roles in skin repair, anti-aging, and wound healing. First identified in human plasma in the 1970s, GHK-Cu levels decline with age, prompting research into its therapeutic applications. As of February 15, 2026, GHK-Cu remains classified primarily as a cosmetic ingredient rather than an FDA-approved drug. No prescription formulations of copper peptide GHK-Cu have received FDA approval for any medical indication, and its use is off-label or investigational in clinical contexts.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, focusing on mechanisms, efficacy, and safety. Targeted PubMed searches yielded 14 high-quality references, including clinical trials and mechanistic studies, supplemented by 4 authoritative sources due to the niche nature of recent systematic reviews on copper peptide GHK-Cu. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on FDA status and long-term safety for this specific query. All claims distinguish FDA-approved (none) from investigational findings. Patients should consult healthcare providers before use, especially with topicals containing metals like copper, due to potential interactions or sensitivities.
Introduction
GHK-Cu, or glycyl-L-histidyl-L-lysine copper, is a naturally occurring tripeptide-copper complex found in human plasma, saliva, and urine, with levels declining with age. It has garnered attention in dermatology and regenerative medicine for its explored roles in wound healing, anti-inflammatory effects, and tissue remodeling [1, 5, 9, 10]. While extensively studied in preclinical and small clinical settings, GHK-Cu lacks FDA approval as a pharmaceutical agent and is primarily available in cosmetic formulations for topical use. No injectable or systemic formulations are FDA-approved for any indication as of February 14, 2026 [11].
This review synthesizes evidence from peer-reviewed journals published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Targeted PubMed searches yielded 8 high-quality peer-reviewed references meeting criteria, falling short of the 12-reference threshold. Thus, primary evidence is supplemented by authoritative sources including NIH.gov (PubChem, ClinicalTrials.gov) and Cleveland Clinic educational materials due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational findings. Readers should consult healthcare providers before use, as data remain preliminary and products vary in quality [10].
Introduction
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.
Introduction
GLP-1 agonists, or glucagon-like peptide-1 receptor agonists, represent a cornerstone of modern pharmacotherapy for type 2 diabetes mellitus (T2DM) and obesity management. These injectable or oral medications are designed to mimic the endogenous GLP-1 hormone, enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety. As of February 18, 2026, the class has expanded with established agents like semaglutide, liraglutide, dulaglutide, and exenatide, alongside dual GLP-1/GIP agonists such as tirzepatide, which are often discussed in GLP-1 contexts due to overlapping mechanisms and indications.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and large-scale clinical trials from PubMed-indexed journals. Key trials like STEP, SURPASS, and SELECT have demonstrated their efficacy in glycemic control (HbA1c reductions of 1.0–2.0%) [1, 3, 4], weight loss (10–20% mean body weight reduction) [2, 3, 4], and cardiovascular risk reduction [4, 7]. FDA-approved indications include T2DM and chronic weight management for adults with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity [13]. Investigational uses in heart failure, NASH, and neurodegeneration are currently being explored [10, 11, nature.com].
All claims are grounded in verifiable data up to 2026. Patients should consult healthcare providers for personalized use, as these medications require monitoring for gastrointestinal (GI) effects, pancreatitis risk, and thyroid concerns. This article addresses high-intent queries on mechanisms, approvals, efficacy, safety, and comparisons to provide an evidence-based overview.
Introduction
GLP-1 meds, or glucagon-like peptide-1 receptor agonists (GLP-1 RAs), represent a cornerstone of modern pharmacotherapy for type 2 diabetes (T2D) and obesity management. These injectable or oral agents mimic the endogenous GLP-1 hormone, promoting insulin secretion, suppressing glucagon, slowing gastric emptying, and reducing appetite. By February 2026, eight GLP-1 RAs have received FDA approval, primarily for glycemic control in T2D and chronic weight management in obesity, with expanding cardiovascular (CV) benefits demonstrated in large-scale trials.
This review synthesizes evidence from over 15 peer-reviewed publications (2020–2026), prioritizing systematic reviews, meta-analyses, and phase 3/4 clinical trials extracted from PubMed. Key agents include semaglutide (Ozempic®, Wegovy®, Rybelsus®), tirzepatide (Mounjaro®, Zepbound®; dual GLP-1/GIP RA often categorized with GLP-1 meds), liraglutide (Victoza®, Saxenda®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and others like lixisenatide (Adlyxin®) and oral semaglutide formulations. FDA-approved indications are clearly distinguished from off-label or investigational uses, such as polycystic ovary syndrome (PCOS) or non-alcoholic steatohepatitis (NASH). Efficacy data show HbA1c reductions of 1.0–2.0% and weight loss of 10–20% body weight, with superior CV risk reduction versus older therapies.
All claims are evidence-based, emphasizing the need for medical supervision due to gastrointestinal (GI) risks, potential thyroid concerns, and contraindications like personal/family history of medullary thyroid carcinoma (MTC). Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to the rapid evolution of post-marketing data on this high-interest topic.
Introduction
AOD9604 is a synthetic peptide derived from the C-terminal fragment (amino acids 177-191) of human growth hormone (hGH), specifically designed to mimic the lipolytic (fat-burning) properties of hGH without its anabolic or hyperglycemic effects. Developed initially by Metabolic Pharmaceuticals in the early 2000s, AOD9604 has garnered interest primarily for potential applications in obesity and fat metabolism. However, despite preclinical promise in animal models, human clinical data remain limited, with no FDA approval for any indication as of February 17, 2026.
Peer-reviewed literature on AOD9604 is sparse, with most studies predating 2010 and focusing on early-phase trials or mechanistic investigations. No systematic reviews, meta-analyses, or large phase 3 trials published between 2020 and 2026 were identified in PubMed searches. Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to limited recent peer-reviewed publications on this specific query. Key findings indicate modest lipolytic effects in vitro and in rodents, but inconsistent weight loss in humans, alongside regulatory classification as an unapproved substance often associated with compounded peptides. This article reviews available evidence, distinguishing FDA-approved contexts (none for AOD9604) from investigational findings, and emphasizes the need for medical supervision. All claims are based on verifiable sources up to the current date.
Introduction
Peptides for muscle growth have gained significant attention in fitness and bodybuilding communities, often promoted as alternatives to anabolic steroids for enhancing hypertrophy, recovery, and performance. These short chains of amino acids mimic natural hormones or growth factors, potentially stimulating muscle protein synthesis, growth hormone (GH) release, or inhibiting myostatin. However, the scientific evidence supporting their use in healthy adults remains limited, with most data derived from preclinical studies, small clinical trials in specific populations (e.g., sarcopenia or HIV-associated wasting), or off-label applications.
This review synthesizes peer-reviewed evidence from 2020–2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Searches yielded only 8 high-quality PubMed-indexed studies directly addressing peptides for muscle growth outcomes, primarily focusing on GH-releasing peptides (GHRPs) like ipamorelin and CJC-1295, or myostatin inhibitors like follistatin. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved indications (none for muscle growth in healthy adults) from investigational or off-label uses. No peptide is FDA-approved for muscle growth or athletic performance enhancement as of February 16, 2026. Users should consult healthcare providers, as self-administration carries risks of contamination, dosing errors, and regulatory violations.
Introduction
Ipamorelin is a synthetic pentapeptide classified as a growth hormone secretagogue (GHS) that selectively stimulates the release of growth hormone (GH) from the pituitary gland. Unlike broader-spectrum GHSs, ipamorelin targets the growth hormone secretagogue receptor (GHSR) with high specificity, with preclinical models showing minimal impact on ACTH or cortisol [peptidedosages.com]. Originally developed in the 1990s, interest in ipamorelin has persisted into 2026 for its potential in age-related GH decline, muscle wasting, and metabolic disorders, though these remain investigational uses. However, as of February 16, 2026, ipamorelin remains investigational and lacks FDA approval for any human therapeutic indication [droracle.ai].
Peer-reviewed literature on ipamorelin from 2020 to 2026 is limited, with only 8 high-quality studies identified via PubMed searches (primarily preclinical or small human trials). Primary evidence is supplemented by authoritative sources, including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic, due to the limited number of recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational/off-label findings. This review synthesizes the latest evidence, emphasizing the need for medical supervision, as self-administration carries risks and legal implications in many jurisdictions. Evidence highlights ipamorelin’s favorable safety profile in short-term studies but underscores gaps in long-term data.
Introduction
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
Introduction
Copper peptide GHK-Cu, also known as glycyl-L-histidyl-L-lysine copper complex or copper tripeptide-1, has garnered significant interest in dermatology and regenerative medicine for its potential roles in skin repair, anti-aging, and wound healing. First identified in human plasma in the 1970s, GHK-Cu levels decline with age, prompting research into its therapeutic applications. As of February 15, 2026, GHK-Cu remains classified primarily as a cosmetic ingredient rather than an FDA-approved drug. No prescription formulations of copper peptide GHK-Cu have received FDA approval for any medical indication, and its use is off-label or investigational in clinical contexts.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, focusing on mechanisms, efficacy, and safety. Targeted PubMed searches yielded 14 high-quality references, including clinical trials and mechanistic studies, supplemented by 4 authoritative sources due to the niche nature of recent systematic reviews on copper peptide GHK-Cu. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on FDA status and long-term safety for this specific query. All claims distinguish FDA-approved (none) from investigational findings. Patients should consult healthcare providers before use, especially with topicals containing metals like copper, due to potential interactions or sensitivities.
Introduction
GHK-Cu, or glycyl-L-histidyl-L-lysine copper, is a naturally occurring tripeptide-copper complex found in human plasma, saliva, and urine, with levels declining with age. It has garnered attention in dermatology and regenerative medicine for its explored roles in wound healing, anti-inflammatory effects, and tissue remodeling [1, 5, 9, 10]. While extensively studied in preclinical and small clinical settings, GHK-Cu lacks FDA approval as a pharmaceutical agent and is primarily available in cosmetic formulations for topical use. No injectable or systemic formulations are FDA-approved for any indication as of February 14, 2026 [11].
This review synthesizes evidence from peer-reviewed journals published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Targeted PubMed searches yielded 8 high-quality peer-reviewed references meeting criteria, falling short of the 12-reference threshold. Thus, primary evidence is supplemented by authoritative sources including NIH.gov (PubChem, ClinicalTrials.gov) and Cleveland Clinic educational materials due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational findings. Readers should consult healthcare providers before use, as data remain preliminary and products vary in quality [10].
Introduction
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.
Introduction
GLP-1 agonists, or glucagon-like peptide-1 receptor agonists, represent a cornerstone of modern pharmacotherapy for type 2 diabetes mellitus (T2DM) and obesity management. These injectable or oral medications are designed to mimic the endogenous GLP-1 hormone, enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety. As of February 18, 2026, the class has expanded with established agents like semaglutide, liraglutide, dulaglutide, and exenatide, alongside dual GLP-1/GIP agonists such as tirzepatide, which are often discussed in GLP-1 contexts due to overlapping mechanisms and indications.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and large-scale clinical trials from PubMed-indexed journals. Key trials like STEP, SURPASS, and SELECT have demonstrated their efficacy in glycemic control (HbA1c reductions of 1.0–2.0%) [1, 3, 4], weight loss (10–20% mean body weight reduction) [2, 3, 4], and cardiovascular risk reduction [4, 7]. FDA-approved indications include T2DM and chronic weight management for adults with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity [13]. Investigational uses in heart failure, NASH, and neurodegeneration are currently being explored [10, 11, nature.com].
All claims are grounded in verifiable data up to 2026. Patients should consult healthcare providers for personalized use, as these medications require monitoring for gastrointestinal (GI) effects, pancreatitis risk, and thyroid concerns. This article addresses high-intent queries on mechanisms, approvals, efficacy, safety, and comparisons to provide an evidence-based overview.
Introduction
GLP-1 meds, or glucagon-like peptide-1 receptor agonists (GLP-1 RAs), represent a cornerstone of modern pharmacotherapy for type 2 diabetes (T2D) and obesity management. These injectable or oral agents mimic the endogenous GLP-1 hormone, promoting insulin secretion, suppressing glucagon, slowing gastric emptying, and reducing appetite. By February 2026, eight GLP-1 RAs have received FDA approval, primarily for glycemic control in T2D and chronic weight management in obesity, with expanding cardiovascular (CV) benefits demonstrated in large-scale trials.
This review synthesizes evidence from over 15 peer-reviewed publications (2020–2026), prioritizing systematic reviews, meta-analyses, and phase 3/4 clinical trials extracted from PubMed. Key agents include semaglutide (Ozempic®, Wegovy®, Rybelsus®), tirzepatide (Mounjaro®, Zepbound®; dual GLP-1/GIP RA often categorized with GLP-1 meds), liraglutide (Victoza®, Saxenda®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and others like lixisenatide (Adlyxin®) and oral semaglutide formulations. FDA-approved indications are clearly distinguished from off-label or investigational uses, such as polycystic ovary syndrome (PCOS) or non-alcoholic steatohepatitis (NASH). Efficacy data show HbA1c reductions of 1.0–2.0% and weight loss of 10–20% body weight, with superior CV risk reduction versus older therapies.
All claims are evidence-based, emphasizing the need for medical supervision due to gastrointestinal (GI) risks, potential thyroid concerns, and contraindications like personal/family history of medullary thyroid carcinoma (MTC). Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to the rapid evolution of post-marketing data on this high-interest topic.
Introduction
AOD9604 is a synthetic peptide derived from the C-terminal fragment (amino acids 177-191) of human growth hormone (hGH), specifically designed to mimic the lipolytic (fat-burning) properties of hGH without its anabolic or hyperglycemic effects. Developed initially by Metabolic Pharmaceuticals in the early 2000s, AOD9604 has garnered interest primarily for potential applications in obesity and fat metabolism. However, despite preclinical promise in animal models, human clinical data remain limited, with no FDA approval for any indication as of February 17, 2026.
Peer-reviewed literature on AOD9604 is sparse, with most studies predating 2010 and focusing on early-phase trials or mechanistic investigations. No systematic reviews, meta-analyses, or large phase 3 trials published between 2020 and 2026 were identified in PubMed searches. Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to limited recent peer-reviewed publications on this specific query. Key findings indicate modest lipolytic effects in vitro and in rodents, but inconsistent weight loss in humans, alongside regulatory classification as an unapproved substance often associated with compounded peptides. This article reviews available evidence, distinguishing FDA-approved contexts (none for AOD9604) from investigational findings, and emphasizes the need for medical supervision. All claims are based on verifiable sources up to the current date.
Introduction
Peptides for muscle growth have gained significant attention in fitness and bodybuilding communities, often promoted as alternatives to anabolic steroids for enhancing hypertrophy, recovery, and performance. These short chains of amino acids mimic natural hormones or growth factors, potentially stimulating muscle protein synthesis, growth hormone (GH) release, or inhibiting myostatin. However, the scientific evidence supporting their use in healthy adults remains limited, with most data derived from preclinical studies, small clinical trials in specific populations (e.g., sarcopenia or HIV-associated wasting), or off-label applications.
This review synthesizes peer-reviewed evidence from 2020–2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Searches yielded only 8 high-quality PubMed-indexed studies directly addressing peptides for muscle growth outcomes, primarily focusing on GH-releasing peptides (GHRPs) like ipamorelin and CJC-1295, or myostatin inhibitors like follistatin. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved indications (none for muscle growth in healthy adults) from investigational or off-label uses. No peptide is FDA-approved for muscle growth or athletic performance enhancement as of February 16, 2026. Users should consult healthcare providers, as self-administration carries risks of contamination, dosing errors, and regulatory violations.
Introduction
Ipamorelin is a synthetic pentapeptide classified as a growth hormone secretagogue (GHS) that selectively stimulates the release of growth hormone (GH) from the pituitary gland. Unlike broader-spectrum GHSs, ipamorelin targets the growth hormone secretagogue receptor (GHSR) with high specificity, with preclinical models showing minimal impact on ACTH or cortisol [peptidedosages.com]. Originally developed in the 1990s, interest in ipamorelin has persisted into 2026 for its potential in age-related GH decline, muscle wasting, and metabolic disorders, though these remain investigational uses. However, as of February 16, 2026, ipamorelin remains investigational and lacks FDA approval for any human therapeutic indication [droracle.ai].
Peer-reviewed literature on ipamorelin from 2020 to 2026 is limited, with only 8 high-quality studies identified via PubMed searches (primarily preclinical or small human trials). Primary evidence is supplemented by authoritative sources, including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic, due to the limited number of recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational/off-label findings. This review synthesizes the latest evidence, emphasizing the need for medical supervision, as self-administration carries risks and legal implications in many jurisdictions. Evidence highlights ipamorelin’s favorable safety profile in short-term studies but underscores gaps in long-term data.
Introduction
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
Introduction
Copper peptide GHK-Cu, also known as glycyl-L-histidyl-L-lysine copper complex or copper tripeptide-1, has garnered significant interest in dermatology and regenerative medicine for its potential roles in skin repair, anti-aging, and wound healing. First identified in human plasma in the 1970s, GHK-Cu levels decline with age, prompting research into its therapeutic applications. As of February 15, 2026, GHK-Cu remains classified primarily as a cosmetic ingredient rather than an FDA-approved drug. No prescription formulations of copper peptide GHK-Cu have received FDA approval for any medical indication, and its use is off-label or investigational in clinical contexts.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, focusing on mechanisms, efficacy, and safety. Targeted PubMed searches yielded 14 high-quality references, including clinical trials and mechanistic studies, supplemented by 4 authoritative sources due to the niche nature of recent systematic reviews on copper peptide GHK-Cu. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on FDA status and long-term safety for this specific query. All claims distinguish FDA-approved (none) from investigational findings. Patients should consult healthcare providers before use, especially with topicals containing metals like copper, due to potential interactions or sensitivities.
Introduction
GHK-Cu, or glycyl-L-histidyl-L-lysine copper, is a naturally occurring tripeptide-copper complex found in human plasma, saliva, and urine, with levels declining with age. It has garnered attention in dermatology and regenerative medicine for its explored roles in wound healing, anti-inflammatory effects, and tissue remodeling [1, 5, 9, 10]. While extensively studied in preclinical and small clinical settings, GHK-Cu lacks FDA approval as a pharmaceutical agent and is primarily available in cosmetic formulations for topical use. No injectable or systemic formulations are FDA-approved for any indication as of February 14, 2026 [11].
This review synthesizes evidence from peer-reviewed journals published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Targeted PubMed searches yielded 8 high-quality peer-reviewed references meeting criteria, falling short of the 12-reference threshold. Thus, primary evidence is supplemented by authoritative sources including NIH.gov (PubChem, ClinicalTrials.gov) and Cleveland Clinic educational materials due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational findings. Readers should consult healthcare providers before use, as data remain preliminary and products vary in quality [10].
Introduction
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.
Introduction
GLP-1 agonists, or glucagon-like peptide-1 receptor agonists, represent a cornerstone of modern pharmacotherapy for type 2 diabetes mellitus (T2DM) and obesity management. These injectable or oral medications are designed to mimic the endogenous GLP-1 hormone, enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety. As of February 18, 2026, the class has expanded with established agents like semaglutide, liraglutide, dulaglutide, and exenatide, alongside dual GLP-1/GIP agonists such as tirzepatide, which are often discussed in GLP-1 contexts due to overlapping mechanisms and indications.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and large-scale clinical trials from PubMed-indexed journals. Key trials like STEP, SURPASS, and SELECT have demonstrated their efficacy in glycemic control (HbA1c reductions of 1.0–2.0%) [1, 3, 4], weight loss (10–20% mean body weight reduction) [2, 3, 4], and cardiovascular risk reduction [4, 7]. FDA-approved indications include T2DM and chronic weight management for adults with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity [13]. Investigational uses in heart failure, NASH, and neurodegeneration are currently being explored [10, 11, nature.com].
All claims are grounded in verifiable data up to 2026. Patients should consult healthcare providers for personalized use, as these medications require monitoring for gastrointestinal (GI) effects, pancreatitis risk, and thyroid concerns. This article addresses high-intent queries on mechanisms, approvals, efficacy, safety, and comparisons to provide an evidence-based overview.
Introduction
GLP-1 meds, or glucagon-like peptide-1 receptor agonists (GLP-1 RAs), represent a cornerstone of modern pharmacotherapy for type 2 diabetes (T2D) and obesity management. These injectable or oral agents mimic the endogenous GLP-1 hormone, promoting insulin secretion, suppressing glucagon, slowing gastric emptying, and reducing appetite. By February 2026, eight GLP-1 RAs have received FDA approval, primarily for glycemic control in T2D and chronic weight management in obesity, with expanding cardiovascular (CV) benefits demonstrated in large-scale trials.
This review synthesizes evidence from over 15 peer-reviewed publications (2020–2026), prioritizing systematic reviews, meta-analyses, and phase 3/4 clinical trials extracted from PubMed. Key agents include semaglutide (Ozempic®, Wegovy®, Rybelsus®), tirzepatide (Mounjaro®, Zepbound®; dual GLP-1/GIP RA often categorized with GLP-1 meds), liraglutide (Victoza®, Saxenda®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and others like lixisenatide (Adlyxin®) and oral semaglutide formulations. FDA-approved indications are clearly distinguished from off-label or investigational uses, such as polycystic ovary syndrome (PCOS) or non-alcoholic steatohepatitis (NASH). Efficacy data show HbA1c reductions of 1.0–2.0% and weight loss of 10–20% body weight, with superior CV risk reduction versus older therapies.
All claims are evidence-based, emphasizing the need for medical supervision due to gastrointestinal (GI) risks, potential thyroid concerns, and contraindications like personal/family history of medullary thyroid carcinoma (MTC). Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to the rapid evolution of post-marketing data on this high-interest topic.
Introduction
AOD9604 is a synthetic peptide derived from the C-terminal fragment (amino acids 177-191) of human growth hormone (hGH), specifically designed to mimic the lipolytic (fat-burning) properties of hGH without its anabolic or hyperglycemic effects. Developed initially by Metabolic Pharmaceuticals in the early 2000s, AOD9604 has garnered interest primarily for potential applications in obesity and fat metabolism. However, despite preclinical promise in animal models, human clinical data remain limited, with no FDA approval for any indication as of February 17, 2026.
Peer-reviewed literature on AOD9604 is sparse, with most studies predating 2010 and focusing on early-phase trials or mechanistic investigations. No systematic reviews, meta-analyses, or large phase 3 trials published between 2020 and 2026 were identified in PubMed searches. Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to limited recent peer-reviewed publications on this specific query. Key findings indicate modest lipolytic effects in vitro and in rodents, but inconsistent weight loss in humans, alongside regulatory classification as an unapproved substance often associated with compounded peptides. This article reviews available evidence, distinguishing FDA-approved contexts (none for AOD9604) from investigational findings, and emphasizes the need for medical supervision. All claims are based on verifiable sources up to the current date.
Introduction
Peptides for muscle growth have gained significant attention in fitness and bodybuilding communities, often promoted as alternatives to anabolic steroids for enhancing hypertrophy, recovery, and performance. These short chains of amino acids mimic natural hormones or growth factors, potentially stimulating muscle protein synthesis, growth hormone (GH) release, or inhibiting myostatin. However, the scientific evidence supporting their use in healthy adults remains limited, with most data derived from preclinical studies, small clinical trials in specific populations (e.g., sarcopenia or HIV-associated wasting), or off-label applications.
This review synthesizes peer-reviewed evidence from 2020–2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Searches yielded only 8 high-quality PubMed-indexed studies directly addressing peptides for muscle growth outcomes, primarily focusing on GH-releasing peptides (GHRPs) like ipamorelin and CJC-1295, or myostatin inhibitors like follistatin. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved indications (none for muscle growth in healthy adults) from investigational or off-label uses. No peptide is FDA-approved for muscle growth or athletic performance enhancement as of February 16, 2026. Users should consult healthcare providers, as self-administration carries risks of contamination, dosing errors, and regulatory violations.
Introduction
Ipamorelin is a synthetic pentapeptide classified as a growth hormone secretagogue (GHS) that selectively stimulates the release of growth hormone (GH) from the pituitary gland. Unlike broader-spectrum GHSs, ipamorelin targets the growth hormone secretagogue receptor (GHSR) with high specificity, with preclinical models showing minimal impact on ACTH or cortisol [peptidedosages.com]. Originally developed in the 1990s, interest in ipamorelin has persisted into 2026 for its potential in age-related GH decline, muscle wasting, and metabolic disorders, though these remain investigational uses. However, as of February 16, 2026, ipamorelin remains investigational and lacks FDA approval for any human therapeutic indication [droracle.ai].
Peer-reviewed literature on ipamorelin from 2020 to 2026 is limited, with only 8 high-quality studies identified via PubMed searches (primarily preclinical or small human trials). Primary evidence is supplemented by authoritative sources, including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic, due to the limited number of recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational/off-label findings. This review synthesizes the latest evidence, emphasizing the need for medical supervision, as self-administration carries risks and legal implications in many jurisdictions. Evidence highlights ipamorelin’s favorable safety profile in short-term studies but underscores gaps in long-term data.
Introduction
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
Introduction
Copper peptide GHK-Cu, also known as glycyl-L-histidyl-L-lysine copper complex or copper tripeptide-1, has garnered significant interest in dermatology and regenerative medicine for its potential roles in skin repair, anti-aging, and wound healing. First identified in human plasma in the 1970s, GHK-Cu levels decline with age, prompting research into its therapeutic applications. As of February 15, 2026, GHK-Cu remains classified primarily as a cosmetic ingredient rather than an FDA-approved drug. No prescription formulations of copper peptide GHK-Cu have received FDA approval for any medical indication, and its use is off-label or investigational in clinical contexts.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, focusing on mechanisms, efficacy, and safety. Targeted PubMed searches yielded 14 high-quality references, including clinical trials and mechanistic studies, supplemented by 4 authoritative sources due to the niche nature of recent systematic reviews on copper peptide GHK-Cu. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on FDA status and long-term safety for this specific query. All claims distinguish FDA-approved (none) from investigational findings. Patients should consult healthcare providers before use, especially with topicals containing metals like copper, due to potential interactions or sensitivities.
Introduction
GHK-Cu, or glycyl-L-histidyl-L-lysine copper, is a naturally occurring tripeptide-copper complex found in human plasma, saliva, and urine, with levels declining with age. It has garnered attention in dermatology and regenerative medicine for its explored roles in wound healing, anti-inflammatory effects, and tissue remodeling [1, 5, 9, 10]. While extensively studied in preclinical and small clinical settings, GHK-Cu lacks FDA approval as a pharmaceutical agent and is primarily available in cosmetic formulations for topical use. No injectable or systemic formulations are FDA-approved for any indication as of February 14, 2026 [11].
This review synthesizes evidence from peer-reviewed journals published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Targeted PubMed searches yielded 8 high-quality peer-reviewed references meeting criteria, falling short of the 12-reference threshold. Thus, primary evidence is supplemented by authoritative sources including NIH.gov (PubChem, ClinicalTrials.gov) and Cleveland Clinic educational materials due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational findings. Readers should consult healthcare providers before use, as data remain preliminary and products vary in quality [10].
Introduction
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.
Introduction
GLP-1 agonists, or glucagon-like peptide-1 receptor agonists, represent a cornerstone of modern pharmacotherapy for type 2 diabetes mellitus (T2DM) and obesity management. These injectable or oral medications are designed to mimic the endogenous GLP-1 hormone, enhancing insulin secretion, suppressing glucagon release, slowing gastric emptying, and promoting satiety. As of February 18, 2026, the class has expanded with established agents like semaglutide, liraglutide, dulaglutide, and exenatide, alongside dual GLP-1/GIP agonists such as tirzepatide, which are often discussed in GLP-1 contexts due to overlapping mechanisms and indications.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and large-scale clinical trials from PubMed-indexed journals. Key trials like STEP, SURPASS, and SELECT have demonstrated their efficacy in glycemic control (HbA1c reductions of 1.0–2.0%) [1, 3, 4], weight loss (10–20% mean body weight reduction) [2, 3, 4], and cardiovascular risk reduction [4, 7]. FDA-approved indications include T2DM and chronic weight management for adults with BMI ≥30 kg/m² or ≥27 kg/m² with comorbidities, when used in conjunction with a reduced-calorie diet and increased physical activity [13]. Investigational uses in heart failure, NASH, and neurodegeneration are currently being explored [10, 11, nature.com].
All claims are grounded in verifiable data up to 2026. Patients should consult healthcare providers for personalized use, as these medications require monitoring for gastrointestinal (GI) effects, pancreatitis risk, and thyroid concerns. This article addresses high-intent queries on mechanisms, approvals, efficacy, safety, and comparisons to provide an evidence-based overview.
Introduction
GLP-1 meds, or glucagon-like peptide-1 receptor agonists (GLP-1 RAs), represent a cornerstone of modern pharmacotherapy for type 2 diabetes (T2D) and obesity management. These injectable or oral agents mimic the endogenous GLP-1 hormone, promoting insulin secretion, suppressing glucagon, slowing gastric emptying, and reducing appetite. By February 2026, eight GLP-1 RAs have received FDA approval, primarily for glycemic control in T2D and chronic weight management in obesity, with expanding cardiovascular (CV) benefits demonstrated in large-scale trials.
This review synthesizes evidence from over 15 peer-reviewed publications (2020–2026), prioritizing systematic reviews, meta-analyses, and phase 3/4 clinical trials extracted from PubMed. Key agents include semaglutide (Ozempic®, Wegovy®, Rybelsus®), tirzepatide (Mounjaro®, Zepbound®; dual GLP-1/GIP RA often categorized with GLP-1 meds), liraglutide (Victoza®, Saxenda®), dulaglutide (Trulicity®), exenatide (Bydureon®, Byetta®), and others like lixisenatide (Adlyxin®) and oral semaglutide formulations. FDA-approved indications are clearly distinguished from off-label or investigational uses, such as polycystic ovary syndrome (PCOS) or non-alcoholic steatohepatitis (NASH). Efficacy data show HbA1c reductions of 1.0–2.0% and weight loss of 10–20% body weight, with superior CV risk reduction versus older therapies.
All claims are evidence-based, emphasizing the need for medical supervision due to gastrointestinal (GI) risks, potential thyroid concerns, and contraindications like personal/family history of medullary thyroid carcinoma (MTC). Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to the rapid evolution of post-marketing data on this high-interest topic.
Introduction
AOD9604 is a synthetic peptide derived from the C-terminal fragment (amino acids 177-191) of human growth hormone (hGH), specifically designed to mimic the lipolytic (fat-burning) properties of hGH without its anabolic or hyperglycemic effects. Developed initially by Metabolic Pharmaceuticals in the early 2000s, AOD9604 has garnered interest primarily for potential applications in obesity and fat metabolism. However, despite preclinical promise in animal models, human clinical data remain limited, with no FDA approval for any indication as of February 17, 2026.
Peer-reviewed literature on AOD9604 is sparse, with most studies predating 2010 and focusing on early-phase trials or mechanistic investigations. No systematic reviews, meta-analyses, or large phase 3 trials published between 2020 and 2026 were identified in PubMed searches. Primary evidence is supplemented by authoritative sources including FDA.gov and NIH.gov due to limited recent peer-reviewed publications on this specific query. Key findings indicate modest lipolytic effects in vitro and in rodents, but inconsistent weight loss in humans, alongside regulatory classification as an unapproved substance often associated with compounded peptides. This article reviews available evidence, distinguishing FDA-approved contexts (none for AOD9604) from investigational findings, and emphasizes the need for medical supervision. All claims are based on verifiable sources up to the current date.
Introduction
Peptides for muscle growth have gained significant attention in fitness and bodybuilding communities, often promoted as alternatives to anabolic steroids for enhancing hypertrophy, recovery, and performance. These short chains of amino acids mimic natural hormones or growth factors, potentially stimulating muscle protein synthesis, growth hormone (GH) release, or inhibiting myostatin. However, the scientific evidence supporting their use in healthy adults remains limited, with most data derived from preclinical studies, small clinical trials in specific populations (e.g., sarcopenia or HIV-associated wasting), or off-label applications.
This review synthesizes peer-reviewed evidence from 2020–2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Searches yielded only 8 high-quality PubMed-indexed studies directly addressing peptides for muscle growth outcomes, primarily focusing on GH-releasing peptides (GHRPs) like ipamorelin and CJC-1295, or myostatin inhibitors like follistatin. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved indications (none for muscle growth in healthy adults) from investigational or off-label uses. No peptide is FDA-approved for muscle growth or athletic performance enhancement as of February 16, 2026. Users should consult healthcare providers, as self-administration carries risks of contamination, dosing errors, and regulatory violations.
Introduction
Ipamorelin is a synthetic pentapeptide classified as a growth hormone secretagogue (GHS) that selectively stimulates the release of growth hormone (GH) from the pituitary gland. Unlike broader-spectrum GHSs, ipamorelin targets the growth hormone secretagogue receptor (GHSR) with high specificity, with preclinical models showing minimal impact on ACTH or cortisol [peptidedosages.com]. Originally developed in the 1990s, interest in ipamorelin has persisted into 2026 for its potential in age-related GH decline, muscle wasting, and metabolic disorders, though these remain investigational uses. However, as of February 16, 2026, ipamorelin remains investigational and lacks FDA approval for any human therapeutic indication [droracle.ai].
Peer-reviewed literature on ipamorelin from 2020 to 2026 is limited, with only 8 high-quality studies identified via PubMed searches (primarily preclinical or small human trials). Primary evidence is supplemented by authoritative sources, including FDA.gov, NIH.gov, Mayo Clinic, and Cleveland Clinic, due to the limited number of recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational/off-label findings. This review synthesizes the latest evidence, emphasizing the need for medical supervision, as self-administration carries risks and legal implications in many jurisdictions. Evidence highlights ipamorelin’s favorable safety profile in short-term studies but underscores gaps in long-term data.
Introduction
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
Introduction
Copper peptide GHK-Cu, also known as glycyl-L-histidyl-L-lysine copper complex or copper tripeptide-1, has garnered significant interest in dermatology and regenerative medicine for its potential roles in skin repair, anti-aging, and wound healing. First identified in human plasma in the 1970s, GHK-Cu levels decline with age, prompting research into its therapeutic applications. As of February 15, 2026, GHK-Cu remains classified primarily as a cosmetic ingredient rather than an FDA-approved drug. No prescription formulations of copper peptide GHK-Cu have received FDA approval for any medical indication, and its use is off-label or investigational in clinical contexts.
This review synthesizes evidence from peer-reviewed studies published between 2020 and 2026, focusing on mechanisms, efficacy, and safety. Targeted PubMed searches yielded 14 high-quality references, including clinical trials and mechanistic studies, supplemented by 4 authoritative sources due to the niche nature of recent systematic reviews on copper peptide GHK-Cu. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on FDA status and long-term safety for this specific query. All claims distinguish FDA-approved (none) from investigational findings. Patients should consult healthcare providers before use, especially with topicals containing metals like copper, due to potential interactions or sensitivities.
Introduction
GHK-Cu, or glycyl-L-histidyl-L-lysine copper, is a naturally occurring tripeptide-copper complex found in human plasma, saliva, and urine, with levels declining with age. It has garnered attention in dermatology and regenerative medicine for its explored roles in wound healing, anti-inflammatory effects, and tissue remodeling [1, 5, 9, 10]. While extensively studied in preclinical and small clinical settings, GHK-Cu lacks FDA approval as a pharmaceutical agent and is primarily available in cosmetic formulations for topical use. No injectable or systemic formulations are FDA-approved for any indication as of February 14, 2026 [11].
This review synthesizes evidence from peer-reviewed journals published between 2020 and 2026, prioritizing systematic reviews, meta-analyses, and clinical trials. Targeted PubMed searches yielded 8 high-quality peer-reviewed references meeting criteria, falling short of the 12-reference threshold. Thus, primary evidence is supplemented by authoritative sources including NIH.gov (PubChem, ClinicalTrials.gov) and Cleveland Clinic educational materials due to limited recent peer-reviewed publications on this specific query. All claims distinguish FDA-approved (none) from investigational findings. Readers should consult healthcare providers before use, as data remain preliminary and products vary in quality [10].
Introduction
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.