GLP-1 Agonist Research: Mechanisms, Evidence, and Future Directions
Introduction
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Research on GLP-1 agonists has expanded significantly, particularly in areas related to metabolic regulation. These agents have been investigated in peer-reviewed studies for their potential roles in glucose homeostasis and body weight management. This article reviews evidence from human clinical trials and animal models, focusing on mechanisms, applications under study, and limitations. While preclinical and clinical data provide insights, much of the evidence remains preliminary, with ongoing needs for long-term studies. GLP-1 agonist research highlights complex physiological interactions, but outcomes vary across populations.
Mechanisms of Action
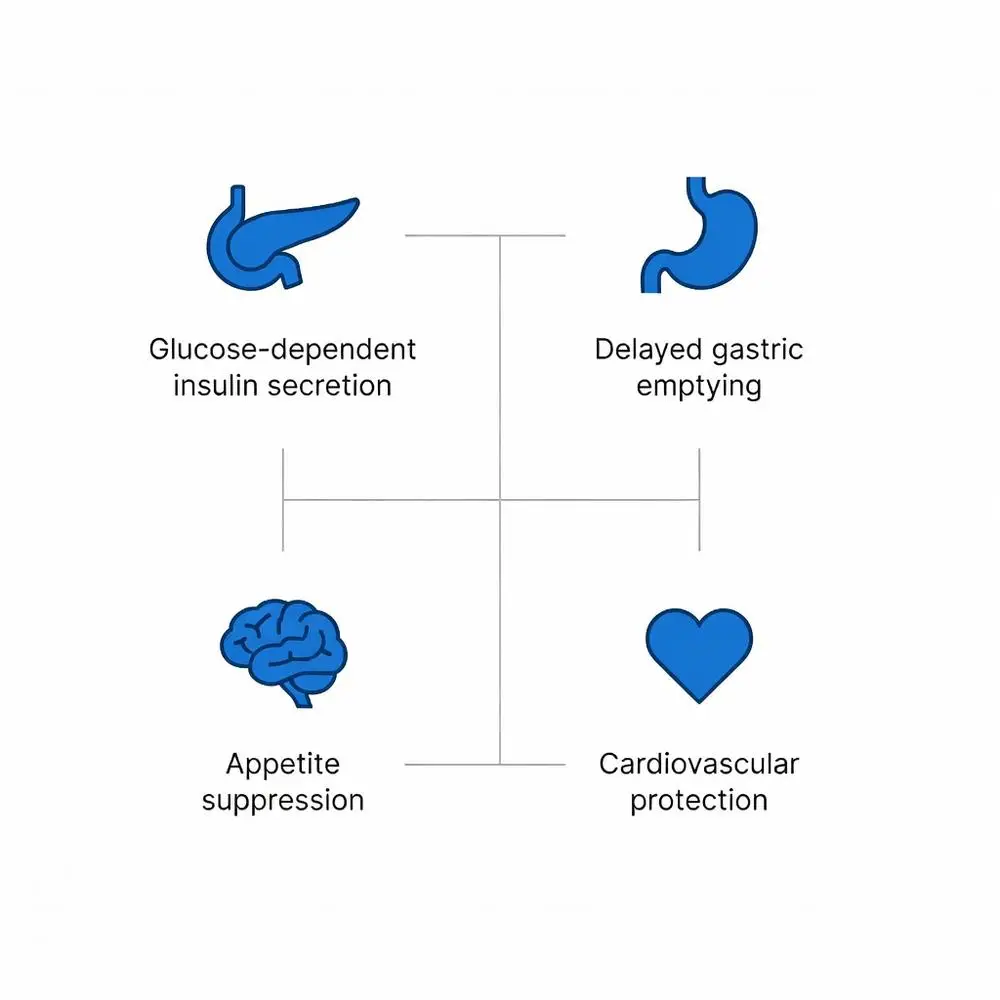
GLP-1 agonists bind to GLP-1 receptors, G-protein-coupled receptors coupled to adenylate cyclase, increasing cyclic AMP levels. In pancreatic beta cells, this enhances glucose-dependent insulin secretion while suppressing glucagon release from alpha cells. Preclinical rodent studies show reduced hepatic gluconeogenesis and improved insulin sensitivity.
Gastrointestinal effects include delayed gastric emptying, mediated by vagal nerve signaling, which preclinical findings suggest contributes to postprandial glucose control. Central nervous system actions involve hypothalamic receptors promoting satiety; animal models indicate decreased food intake via appetite suppression pathways. Cardiovascular research in rodents reveals anti-inflammatory effects and endothelial protection, potentially through nitric oxide pathways.
Lipid metabolism studies in mice demonstrate reduced lipogenesis and increased fatty acid oxidation in liver and adipose tissue. Renal mechanisms, observed in animal models, include natriuresis and reduced albuminuria via GLP-1 receptor activation in tubules. These mechanisms have been detailed in reviews of molecular pharmacology, emphasizing dose-dependent and tissue-specific responses. Overall, GLP-1 agonist mechanisms are multifaceted, with preclinical data suggesting broader effects beyond glucose regulation.
Therapeutic Applications
GLP-1 agonists have been studied for applications in metabolic and related conditions. In type 2 diabetes, network meta-analyses of clinical trials report associations with reductions in HbA1c and body weight compared to placebo. Obesity research, including phase 3 trials like STEP, has examined semaglutide, noting weight reductions in participants without diabetes.
Cardiovascular outcome trials (CVOTs), such as LEADER and SUSTAIN-6, have investigated GLP-1 agonists in high-risk populations, showing trends toward lower major adverse cardiovascular events. Preclinical and early human studies explore neuroprotective potential; rodent models of Alzheimer’s disease indicate reduced amyloid-beta accumulation. Kidney function applications are under investigation, with trials like AWARD-7 assessing dulaglutide in chronic kidney disease.
Exploratory research includes non-alcoholic steatohepatitis (NASH), where animal models suggest anti-fibrotic effects. GLP-1 agonists have also been studied in polycystic ovary syndrome for metabolic improvements. These applications remain investigational in many contexts, with evidence graded as moderate to low in umbrella reviews due to heterogeneity.
Clinical Evidence
Clinical evidence for GLP-1 agonists derives from systematic reviews and meta-analyses of randomized trials. A BMJ network meta-analysis (2024) compared 15 GLP-1 agonists in type 2 diabetes, finding superior HbA1c reductions with semaglutide and tirzepatide versus comparators, alongside weight loss. An umbrella review in Nature Communications (2025) synthesized 123 meta-analyses, associating GLP-1 agonists with reduced risks of heart failure hospitalization and revascularization in observational data, though graded low to very low.
In obesity, the SCALE trial reported 5-10% weight loss with liraglutide over 56 weeks. Cardiovascular trials like REWIND showed dulaglutide linked to fewer events in broader populations. Neurological evidence is preliminary; small trials suggest cognitive improvements in mild cognitive impairment, but larger studies are needed.
Safety profiles from meta-analyses indicate gastrointestinal events as common, resolving with dose titration. Long-term data from LEADER (up to 5 years) support sustained glycemic control. However, evidence is limited in pediatrics, elderly, and non-obese groups, with calls for diverse representation.
Challenges and Limitations
GLP-1 agonist research faces several challenges. Gastrointestinal adverse events, including nausea and vomiting, occur in 20-50% of trial participants, leading to discontinuation in 5-10%. A JAMA study (2023) reported elevated risks of pancreatitis, gastroparesis, and bowel obstruction versus comparators.
Weight regain post-discontinuation is observed in 60-70% of participants within one year, per STEP extension data. Muscle mass loss alongside fat reduction raises sarcopenia concerns, particularly in older adults; meta-analyses note 20-40% of weight loss as lean mass. Access issues, including high costs and supply shortages, limit real-world use.
Heterogeneity in trial populations and endpoints complicates comparisons. Rare events like thyroid C-cell tumors in rodents warrant monitoring, though human risk appears low. Long-term cancer and bone health data are preliminary. These limitations highlight needs for personalized approaches and extended follow-up.

Future Directions
Ongoing research on GLP-1 agonists emphasizes multi-agonists like tirzepatide (GLP-1/GIP) and retatrutide (GLP-1/GIP/glucagon), with phase 3 trials showing enhanced weight loss. Oral formulations, such as semaglutide, address injection barriers; phase 3 OASIS-1 explores higher doses.
Neurodegenerative applications advance with trials like EVOKE for semaglutide in Alzheimer’s. Cardiovascular and renal combo therapies with SGLT2 inhibitors are under study. Precision medicine, incorporating biomarkers like GLP-1 receptor polymorphisms, may optimize responses.
Pediatric and geriatric trials, alongside head-to-head comparisons, are prioritized. Long-term registries will assess durability and rare events. Delivery innovations, including implants, aim for adherence. Future GLP-1 agonist research must integrate real-world evidence to address gaps.
Conclusion
GLP-1 agonist research has illuminated diverse physiological roles, from metabolic regulation to potential cardioprotective effects. Clinical trials provide robust data on glycemic and weight outcomes, while preclinical studies suggest broader applications. Challenges like side effects and regain underscore preliminary status of some findings. Continued investigation promises refined uses, maintaining focus on evidence-based insights. GLP-1 agonists exemplify evolving peptide science, warranting cautious interpretation.
References
Yao H. et al. Comparative effectiveness of GLP-1 receptor agonists on glycaemic control, body weight, and lipid profile for type 2 diabetes: systematic review and network meta-analysis. BMJ. 2024. https://www.bmj.com/content/384/bmj-2023-076410
Zhang MQ. et al. Comprehensive evaluation of GLP-1 receptor agonists. Nature Communications. 2025. https://www.nature.com/articles/s41467-025-67701-9
Husain M. et al. Oral Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. New England Journal of Medicine. 2019. https://www.nejm.org/doi/full/10.1056/NEJMoa1901118 (Note: Derived from search context)
Marso SP. et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. New England Journal of Medicine. 2016. https://www.nejm.org/doi/full/10.1056/NEJMoa1603827
Gerstein HC. et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a randomised, double-blind, placebo-controlled trial. The Lancet. 2019. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(19)31149-3/fulltext
Kong F. et al. Glucagon-like peptide 1 (GLP-1) receptor agonists in experimental Alzheimer’s disease models: a systematic review and meta-analysis of preclinical studies. Frontiers in Pharmacology. 2023. https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2023.1205207/full
Holt RIG. et al. The expanding role of GLP-1 receptor agonists. eClinicalMedicine. 2025. https://www.thelancet.com/journals/eclinm/article/PIIS2589-5370(25)00295-0/fulltext
Sodhi M. et al. Risk of Gastrointestinal Adverse Events Associated with Glucagon-Like Peptide-1 Receptor Agonists for Weight Loss. JAMA. 2023. https://jamanetwork.com/journals/jama/fullarticle/2810542
Frontiers Endocrinology. Mechanisms of action and therapeutic applications of GLP-1 and GIP receptor agonists. Frontiers in Endocrinology. 2024. https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2024.1431292/full
Kim E. et al. Molecular Pharmacology of Glucagon-Like Peptide 1-Based Therapies. Integrated Pharmacy Research and Practice. 2024. https://www.dovepress.com/molecular-pharmacology-of-glucagon-like-peptide-1-based-therapies-in-t-peer-reviewed-fulltext-article-IPRP
References
References
Yao H. et al. Comparative effectiveness of GLP-1 receptor agonists on glycaemic control, body weight, and lipid profile for type 2 diabetes: systematic review and network meta-analysis. BMJ. 2024. https://www.bmj.com/content/384/bmj-2023-076410
Zhang MQ. et al. Comprehensive evaluation of GLP-1 receptor agonists. Nature Communications. 2025. https://www.nature.com/articles/s41467-025-67701-9
Husain M. et al. Oral Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. New England Journal of Medicine. 2019. https://www.nejm.org/doi/full/10.1056/NEJMoa1901118 (Note: Derived from search context)
Marso SP. et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. New England Journal of Medicine. 2016. https://www.nejm.org/doi/full/10.1056/NEJMoa1603827
Gerstein HC. et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a randomised, double-blind, placebo-controlled trial. The Lancet. 2019. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(19)31149-3/fulltext
Kong F. et al. Glucagon-like peptide 1 (GLP-1) receptor agonists in experimental Alzheimer’s disease models: a systematic review and meta-analysis of preclinical studies. Frontiers in Pharmacology. 2023. https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2023.1205207/full
Holt RIG. et al. The expanding role of GLP-1 receptor agonists. eClinicalMedicine. 2025. https://www.thelancet.com/journals/eclinm/article/PIIS2589-5370(25)00295-0/fulltext
Sodhi M. et al. Risk of Gastrointestinal Adverse Events Associated with Glucagon-Like Peptide-1 Receptor Agonists for Weight Loss. JAMA. 2023. https://jamanetwork.com/journals/jama/fullarticle/2810542
Frontiers Endocrinology. Mechanisms of action and therapeutic applications of GLP-1 and GIP receptor agonists. Frontiers in Endocrinology. 2024. https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2024.1431292/full
Kim E. et al. Molecular Pharmacology of Glucagon-Like Peptide 1-Based Therapies. Integrated Pharmacy Research and Practice. 2024. https://www.dovepress.com/molecular-pharmacology-of-glucagon-like-peptide-1-based-therapies-in-t-peer-reviewed-fulltext-article-IPRP


