GLP-1 Agonist Research: Mechanisms, Evidence, and Future Directions
Introduction
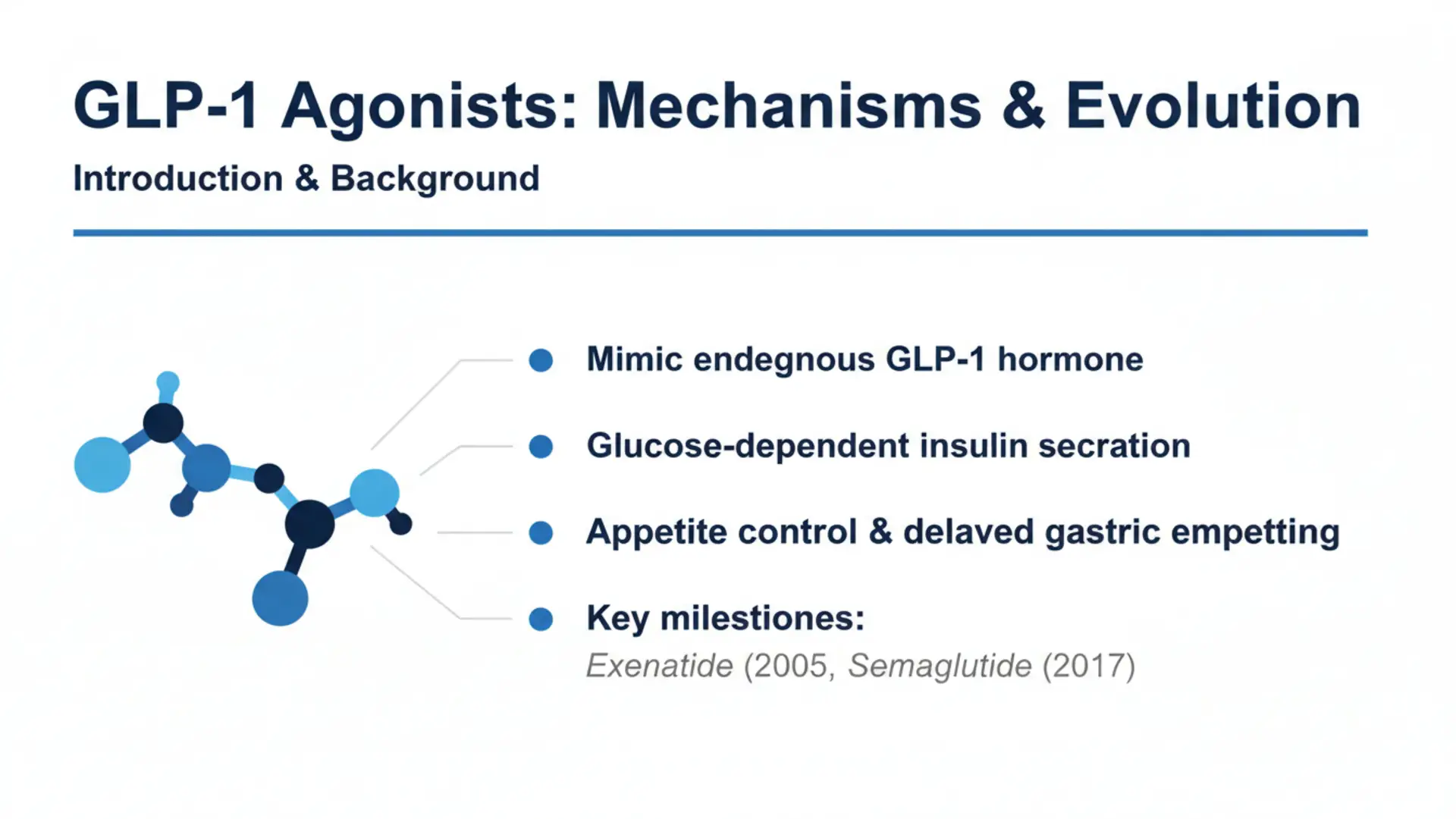
GLP-1 agonists, also known as glucagon-like peptide-1 receptor agonists, represent a class of peptides that mimic the actions of the endogenous incretin hormone GLP-1. Derived from the proglucagon gene, GLP-1 is secreted by intestinal L-cells in response to nutrient ingestion. Research on GLP-1 agonists has expanded significantly since their development in the early 2000s, driven by preclinical and clinical studies exploring their physiological effects. These compounds have been investigated in contexts such as glucose regulation, appetite control, and metabolic processes. Initial discoveries in the 1980s identified GLP-1’s insulinotropic properties, leading to the approval of the first GLP-1 agonist, exenatide, in 2005. Subsequent advancements include long-acting formulations like liraglutide and semaglutide. This article reviews peer-reviewed evidence on GLP-1 agonists, focusing on mechanisms, applications under study, and limitations, while emphasizing that findings are preliminary and evidence varies in strength.

Mechanisms of Action
GLP-1 agonists bind to GLP-1 receptors, G-protein-coupled receptors expressed in pancreatic beta cells, alpha cells, brain, heart, and vasculature. In preclinical models, they enhance glucose-stimulated insulin secretion while suppressing glucagon release under hyperglycemic conditions. Animal studies show delayed gastric emptying via vagal nerve signaling, reducing postprandial glucose excursions. Central nervous system effects include activation of hypothalamic neurons, decreasing food intake in rodents. Cardiovascular research in animal models suggests anti-inflammatory actions, reduced oxidative stress, and improved endothelial function. Preclinical findings indicate modulation of autophagy and endoplasmic reticulum stress in metabolic tissues. Human islet studies confirm preserved insulinotropic effects despite DPP-4 activity. Multi-receptor agonists, combining GLP-1 with GIP activity, amplify these pathways in rodent models. Overall, mechanisms involve cAMP elevation, PKA activation, and downstream effects on ion channels and gene expression, with evidence primarily from cellular and animal investigations.
Therapeutic Applications
GLP-1 agonists have been studied for applications in metabolic and related areas. In type 2 diabetes models, they promote glycemic control through incretin mimicry. Obesity research in humans and animals explores appetite suppression and weight reduction mechanisms. Preclinical studies suggest benefits in non-alcoholic fatty liver disease (NAFLD), with reductions in hepatic steatosis and inflammation in rodent models. Cardiovascular investigations include heart failure and atherosclerosis, where animal data show cardioprotective effects via improved perfusion. Neurological preclinical findings indicate neuroprotection in Alzheimer’s disease models, with decreased amyloid-beta burden and enhanced synaptic plasticity. Substance use disorder research in animals demonstrates reduced intake of alcohol and drugs via reward pathway modulation. Renal studies in diabetic models point to glomerular protection. Emerging areas include polycystic ovary syndrome and sleep apnea, based on observational human data. Evidence is predominantly preclinical or from short-term trials, with no established preventive roles.
Clinical Evidence
Clinical trials provide evidence on GLP-1 agonists primarily in type 2 diabetes and obesity cohorts. Cardiovascular outcome trials like LEADER (liraglutide) and SUSTAIN (semaglutide) reported associations with reduced major adverse events in high-risk patients, graded as high-quality in meta-analyses. Network meta-analyses of randomized trials show dose-dependent reductions in HbA1c and body weight across agents like dulaglutide and tirzepatide. Umbrella reviews of 123 meta-analyses covering 464 outcomes graded 47% as high evidence for metabolic effects, with moderate support for weight management. In obesity trials like STEP, semaglutide linked to sustained weight loss in non-diabetic overweight individuals. NAFLD human studies show histological improvements in small cohorts. Neurological pilots suggest cognitive enhancements in mild impairment, but data are preliminary. Renal outcomes in trials like AWARD-7 indicate albuminuria reductions. Real-world studies note high discontinuation rates (20-50%). Evidence strength varies, with stronger support for glycemic metrics and weaker for extrametabolic outcomes.
Challenges and Limitations
Research on GLP-1 agonists highlights gastrointestinal adverse events as primary concerns, including nausea, vomiting, and diarrhea, reported in up to 40% of trial participants. Cohort studies associate GLP-1 agonists with elevated risks of pancreatitis, gastroparesis, and bowel obstruction compared to alternatives. Rare events like non-arteritic anterior ischemic optic neuropathy appear in pharmacovigilance data. Muscle mass loss during weight reduction poses challenges, observed in obesity trials. High costs and injection requirements limit accessibility, with real-world persistence below 50% at one year. Heterogeneity in formulations complicates comparisons, and long-term safety beyond five years lacks data. Preclinical-to-human translation varies, with animal models overestimating neuroprotective effects. Contraindications include medullary thyroid carcinoma history. Meta-analyses grade many outcomes as low or very low due to observational biases. These limitations underscore the need for cautious interpretation.
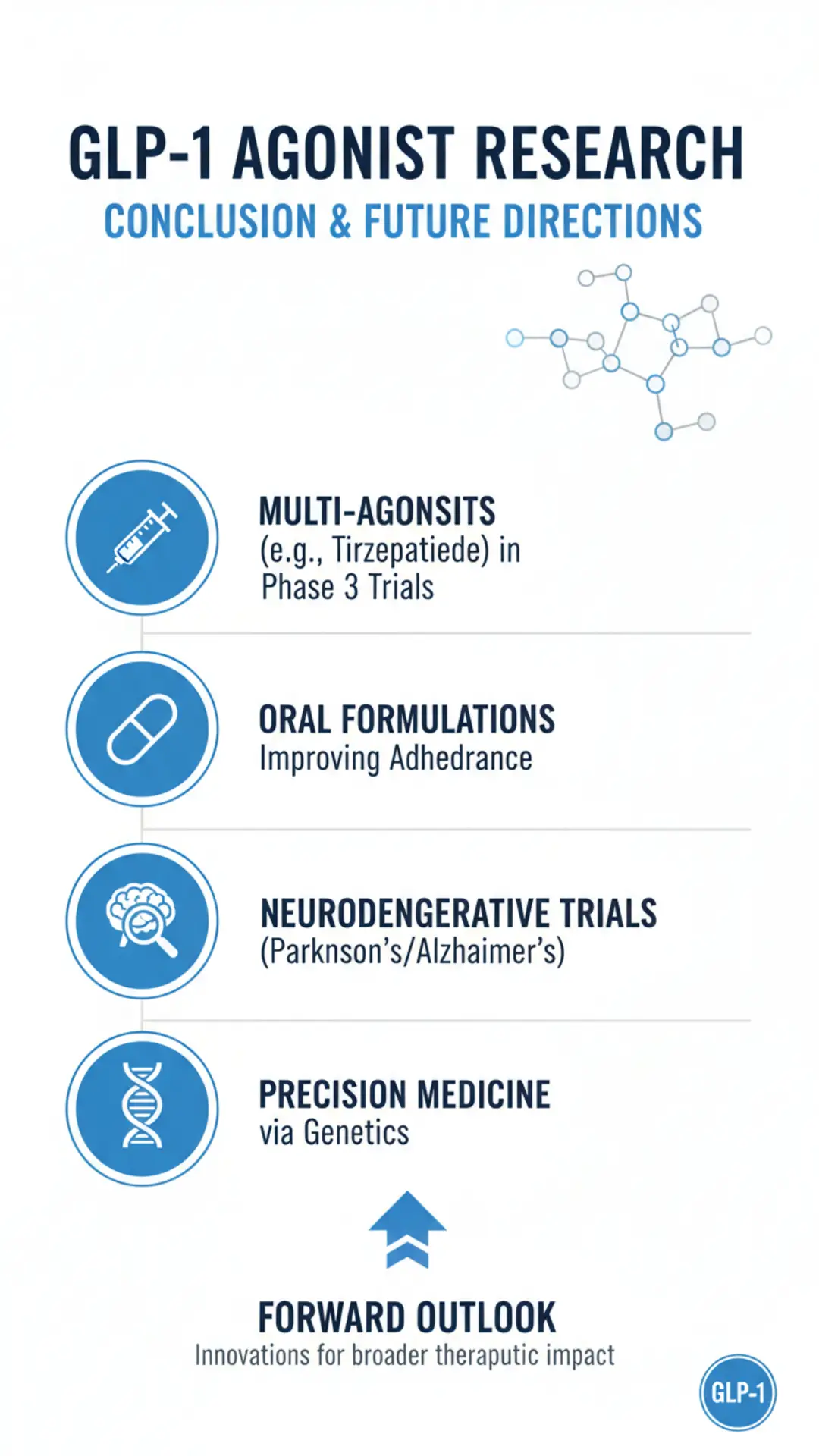
Future Directions
Ongoing research on GLP-1 agonists focuses on multi-agonists like tirzepatide (GLP-1/GIP) and retatrutide (GLP-1/GIP/glucagon), with phase 3 trials showing amplified metabolic effects. Oral formulations address adherence barriers, as seen in oral semaglutide studies. Neurodegenerative trials target Parkinson’s and Alzheimer’s, building on pilot cognitive data. Cardiovascular expansions include heart failure with preserved ejection fraction. Combination therapies with SGLT2 inhibitors are under evaluation for synergistic renal benefits. Pediatric and peripartum applications require safety data. Real-world evidence platforms track long-term outcomes amid rising use. Preclinical work explores small-molecule agonists for non-injectable options. Precision medicine approaches aim to predict responders via genetics. Challenges like supply shortages drive biosimilar development. Future trials emphasize diverse populations and sustained outcomes post-discontinuation.
Conclusion
GLP-1 agonist research has illuminated diverse physiological pathways, from incretin effects to potential metabolic and neuroprotective roles. Preclinical and clinical evidence supports investigations into glucose regulation and weight management, with cardiovascular associations in select trials. However, gastrointestinal risks, limited long-term data, and variable efficacy highlight research gaps. Future studies on multi-agonists and novel indications may expand understanding, but evidence remains preliminary. Continued peer-reviewed scrutiny is essential for informed scientific discourse on GLP-1 agonists.
References
Müller TD, et al. GLP-1 receptor agonists in the treatment of type 2 diabetes – state-of-the-art. Molecular Metabolism. 2020. https://pubmed.ncbi.nlm.nih.gov/33068776/
Wharton S, et al. Emerging Role of GLP-1 Agonists in Obesity. Journal of Obesity & Metabolic Syndrome. 2023. https://pmc.ncbi.nlm.nih.gov/articles/PMC10341852/
Li X, et al. The benefits of GLP1 receptors in cardiovascular diseases. European Journal of Medical Research. 2023. https://pmc.ncbi.nlm.nih.gov/articles/PMC10739421/
Wang Y, et al. GLP-1 receptor agonists: exploration of transformation from metabolic to anti-inflammatory drugs. Frontiers in Immunology. 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC12461214/
Campbell JE. Mechanisms of Action and Therapeutic Application of Glucagon-like Peptide-1. Cell Metabolism. 2018. https://pubmed.ncbi.nlm.nih.gov/29617641/
Zhang H, et al. Recent Advances and Therapeutic Benefits of Glucagon-Like Peptide-1 Agonists. Cureus. 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC11579408/
Palladino R, et al. Anti-inflammatory role of glucagon-like peptide 1 receptor agonists in neurological disorders. Frontiers in Neurology. 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC10823863/
Yao H, et al. Mechanisms of action and therapeutic applications of GLP-1 receptor agonists in diabetes. Pharmaceutics. 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC11304055/
Khalil-Kalantar Z, et al. Animal studies on glucagon-like peptide-1 receptor agonists and non-alcoholic fatty liver disease. eClinicalMedicine. 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC11519281/
Wang C, et al. Comprehensive evaluation of GLP-1 receptor agonists. Nature Communications. 2025. https://www.nature.com/articles/s41467-025-67701-9
References
References
Müller TD, et al. GLP-1 receptor agonists in the treatment of type 2 diabetes – state-of-the-art. Molecular Metabolism. 2020. https://pubmed.ncbi.nlm.nih.gov/33068776/
Wharton S, et al. Emerging Role of GLP-1 Agonists in Obesity. Journal of Obesity & Metabolic Syndrome. 2023. https://pmc.ncbi.nlm.nih.gov/articles/PMC10341852/
Li X, et al. The benefits of GLP1 receptors in cardiovascular diseases. European Journal of Medical Research. 2023. https://pmc.ncbi.nlm.nih.gov/articles/PMC10739421/
Wang Y, et al. GLP-1 receptor agonists: exploration of transformation from metabolic to anti-inflammatory drugs. Frontiers in Immunology. 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC12461214/
Campbell JE. Mechanisms of Action and Therapeutic Application of Glucagon-like Peptide-1. Cell Metabolism. 2018. https://pubmed.ncbi.nlm.nih.gov/29617641/
Zhang H, et al. Recent Advances and Therapeutic Benefits of Glucagon-Like Peptide-1 Agonists. Cureus. 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC11579408/
Palladino R, et al. Anti-inflammatory role of glucagon-like peptide 1 receptor agonists in neurological disorders. Frontiers in Neurology. 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC10823863/
Yao H, et al. Mechanisms of action and therapeutic applications of GLP-1 receptor agonists in diabetes. Pharmaceutics. 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC11304055/
Khalil-Kalantar Z, et al. Animal studies on glucagon-like peptide-1 receptor agonists and non-alcoholic fatty liver disease. eClinicalMedicine. 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC11519281/
Wang C, et al. Comprehensive evaluation of GLP-1 receptor agonists. Nature Communications. 2025. https://www.nature.com/articles/s41467-025-67701-9


