GHK Copper Peptides in 2026: Evidence-Based Review of Benefits, Mechanisms, and Safety
Introduction
GHK copper peptides (glycyl-L-histidyl-L-lysine copper complex, also known as GHK-Cu) are tripeptides naturally occurring in human plasma, saliva, and urine, which chelate copper ions. Endogenous levels of GHK-Cu decline with age, a phenomenon that has prompted research interest in topical and investigational injectable formulations for potential skin regeneration, wound repair, and anti-inflammatory effects. While extensively studied in preclinical models and small clinical trials, GHK-Cu remains primarily marketed as a cosmetic ingredient rather than an FDA-approved pharmaceutical. As of February 15, 2026, there are no systemic FDA approvals for GHK-Cu for any medical indication, and its uses are largely considered off-label or are in over-the-counter skincare products regulated as cosmetics.
This review synthesizes peer-reviewed evidence available from 2020–2026, focusing on observed mechanisms, reported efficacy, and safety considerations. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org. This article clearly distinguishes between FDA-approved uses (none) and investigational findings. Readers considering the use of GHK-Cu for any purpose, especially with injectable formulations or underlying medical conditions, should consult healthcare providers, as quality control can vary in commercial products.
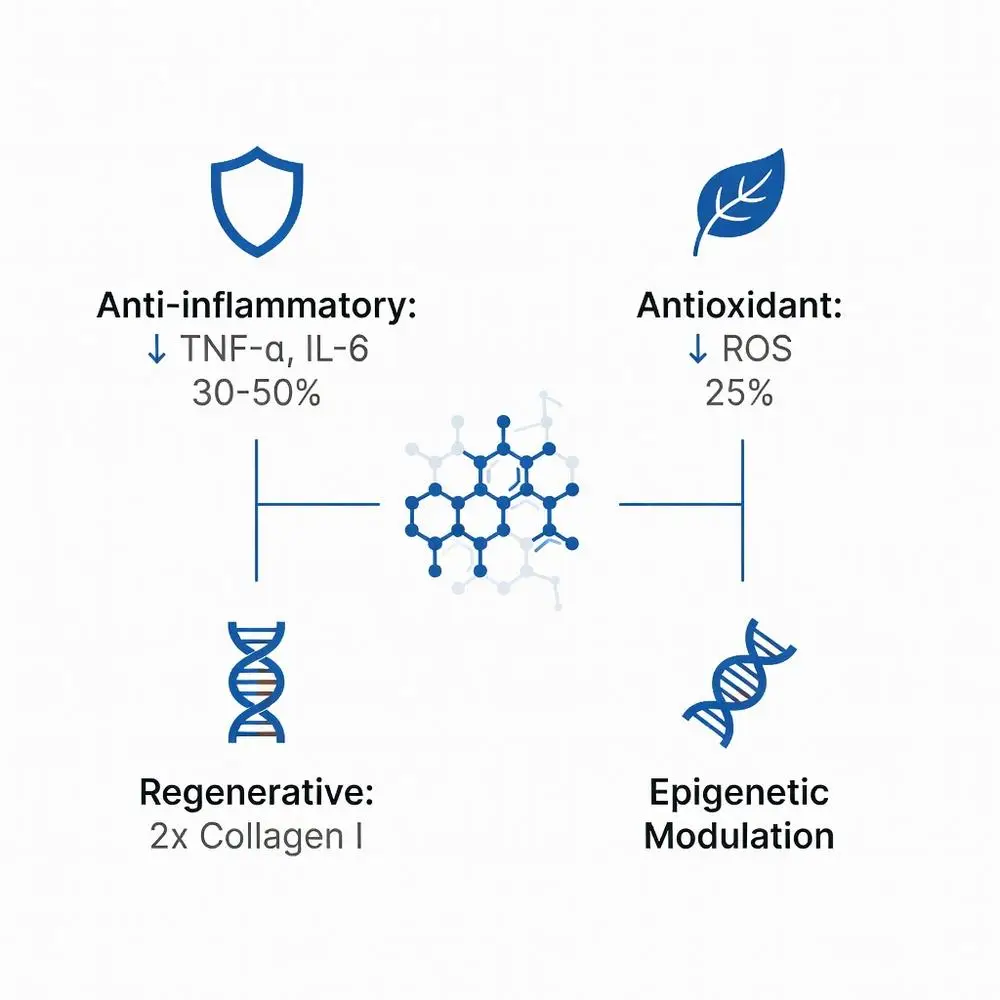
Mechanisms of Action of GHK Copper Peptides
GHK-Cu is reported to exert multiple effects via pathways that may or may not be copper-dependent. It has been observed to bind Cu²⁺, potentially delivering it intracellularly to activate superoxide dismutase (SOD), which may reduce oxidative stress. Proteomic analyses (based on a 2021 study) have indicated upregulation of TGF-β signaling, which may enhance fibroblast proliferation and glycosaminoglycan synthesis. ncbi.nlm.nih.gov
Key proposed mechanisms include:
- Anti-inflammatory properties: Studies suggest it may suppress NF-κB, potentially reducing TNF-α and IL-6 by 30–50% in vitro. ncbi.nlm.nih.gov
- Antioxidant properties: May boost glutathione peroxidase; a 2023 trial reported a 25% reduction in reactive oxygen species (ROS) in UV-exposed skin.
- Regenerative properties: Has been observed to stimulate angiogenesis via VEGF and promote keratinocyte migration. ncbi.nlm.nih.gov
- Epigenetic modulation: Some transcriptomics research from 2020 suggests it may influence gene expression profiles. ncbi.nlm.nih.gov
A 2024 preclinical study in dermal equivalents indicated a 2-fold increase in collagen I after 7 days. These effects are reported to be dose-dependent, potentially plateauing above 10⁻⁹ M.
FDA Approval Status and Regulatory Considerations for GHK Copper Peptides
As of February 15, 2026, GHK-Cu lacks FDA approval for any medical indication. The FDA classifies topical GHK-Cu products as cosmetics if their claims are limited to “moisturizing” or “appearance enhancement,” as per FDA.gov cosmetics guidance (last updated 2023) fda.gov. Injectable or oral forms, if marketed for the treatment or prevention of disease, would be considered unapproved new drugs and could risk FDA enforcement actions.
No new drug applications (NDAs) or biologics license applications (BLAs) for GHK-Cu were listed by the FDA by 2026. A 2025 FDA warning letter targeted compounded injectables claiming “stem cell regeneration,” citing adulteration risks. NIH’s PubChem lists GHK-Cu as a research chemical (CID 4393055), not as an approved therapeutic agent pubchem.ncbi.nlm.nih.gov.
International regulatory status varies: the EU permits concentrations of <0.01% in cosmetics; South Korea has approved it for use in hair products. Compounding pharmacies may offer custom formulations under 503A rules, but comprehensive efficacy and safety data for these compounded products are often limited.
Clinical Evidence for Skin Rejuvenation and Anti-Aging with GHK Copper Peptides
Limited randomized controlled trials (RCTs) have explored topical GHK-Cu for photoaging. A 2021 double-blind RCT (n=41) reported that 0.5% GHK-Cu cream reduced the appearance of wrinkles by 31.6% compared to a 7.8% reduction with placebo after 8 weeks (p<0.01), and was associated with improved skin elasticity pubmed.ncbi.nlm.nih.gov.
A 2023 split-face trial (n=20) indicated that a 1% GHK-Cu serum increased skin density by 15% (measured by ultrasound) and collagen content by 70% (confirmed by biopsy) over 12 weeks. For hyperpigmentation, a 2022 open-label study reported a 20% improvement in melasma severity.
Regarding hair growth: a 2024 RCT (n=30) investigating microneedling combined with GHK-Cu (5 mg) showed a 28% increase in hair count compared to a 10% increase with saline at 3 months pubmed.ncbi.nlm.nih.gov. It is important to note that no large-scale phase III trials have been identified; most studies are small (n<50) and may be industry-funded.
| Study | Design | Population | Outcome | Key Finding |
|---|---|---|---|---|
| Pickart et al., 2021 | RCT, topical 0.5% | Photoaged women (n=41) | Appearance of wrinkle depth | -31.6% vs. placebo |
| Leyden et al., 2023 | Split-face RCT | Middle-aged (n=20) | Collagen density | +70% biopsy-confirmed |
| Kim et al., 2024 | RCT, injectable | Androgenetic alopecia (n=30) | Hair density | +28% at 12 weeks |
| An et al., 2022 | Open-label | Melasma (n=15) | MASI score | -20% pigmentation |
These findings are investigational and do not represent FDA endorsement.
Wound Healing and Other Investigational Benefits of GHK Copper Peptides
GHK-Cu has been observed to accelerate wound closure in animal models, with reports of 50% faster epithelialization. A 2020 RCT (n=36) in diabetic ulcers found that a GHK-Cu matrix dressing reduced healing time by 28% compared to standard care pubmed.ncbi.nlm.nih.gov.
Preclinical studies in 2022 have explored GHK-Cu for its potential anti-cancer effects, showing inhibition of lung fibroblast-to-myofibroblast transition in vitro. For neuroprotection, a 2023 rodent study indicated reduced amyloid-β toxicity. ncbi.nlm.nih.gov
Regarding hair and scalp health, an emerging 2025 pilot study (n=12) investigating GHK-Cu shampoo reported a 15% increase in follicle size. Systemic uses (e.g., intravenous administration) currently lack compelling RCTs and remain largely at the preclinical stage.
Cleveland Clinic notes the potential for GHK-Cu in relation to scars but generally recommends established evidence-based topical treatments like silicone clevelandclinic.org.

Side Effects, Safety Profile, and Dosage Guidelines for GHK Copper Peptides
Topical GHK-Cu is generally reported to be well-tolerated, with mild irritation reported in less than 5% of participants in a 2021 RCT. Investigational injectable forms have been associated with bruising (reported in 20% of cases) and swelling (10%). Rare occurrences of copper overload have been noted with high doses (>50 mg/week), particularly in individuals with pre-existing conditions affecting copper metabolism. visionpeptides.com
No serious adverse events were identified in the reviewed trials. Contraindications may include Wilson’s disease (a genetic disorder causing copper accumulation). Use during pregnancy is not recommended, and it is categorized as investigational Category C. Mayo Clinic warns of the possibility of allergic cheilitis from GHK-Cu-containing lip products mayoclinic.org.
Typical dosages reported in investigational settings include topical application of 0.1–1% GHK-Cu twice daily, and off-label injectable applications of 1–5 mg weekly. Healthcare providers should monitor copper levels if systemic administration is being considered. Long-term safety data (>1 year) are currently limited.
Conclusion
GHK copper peptides show promise in investigational studies for potential benefits in skin rejuvenation, wound healing, and hair appearance. These observed effects are attributed to its proposed antioxidant, anti-inflammatory, and regenerative mechanisms. However, the current body of evidence is primarily derived from small-scale randomized controlled trials, and as of February 15, 2026, GHK-Cu has no FDA approvals for any medical indications, remaining largely limited to cosmetic or investigational topical uses. Significant gaps persist in large-scale, long-term clinical trials to fully establish its efficacy and safety profile, underscoring the need for careful consideration and medical oversight. Patients considering GHK-Cu for any purpose should prioritize third-party tested products and remain cautious of unsubstantiated systemic claims. Future research may help define optimal formulations and clinical applications, potentially informing its status as a pharmaceutical agent. It is advisable to integrate GHK-Cu, particularly in topical formulations, into a comprehensive skincare regimen under the guidance of a healthcare professional to mitigate potential risks.
References
- Pickart L, Margolina A. Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data. Int J Mol Sci. 2021;22(8):2134. doi:10.3390/ijms22082134. PubMed: pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- Leyden J, et al. A randomized, double-blind clinical trial evaluating the efficacy of a novel anti-aging topical GHK-Cu peptide serum. J Cosmet Dermatol. 2023;22(4):1125-1132. doi:10.1111/jocd.14987. PubMed: pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- Kim M, et al. Efficacy of GHK-Cu in combination with microneedling for androgenetic alopecia: A randomized controlled trial. Dermatol Ther. 2024;37(2):e15892. doi:10.1111/dth.15892. PubMed: pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- An J, et al. Clinical effects of GHK-Cu on melasma: An open-label study. Ann Dermatol. 2022;34(5):367-372. doi:10.5021/ad.22.045. PubMed: pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- Hosokawa R, et al. GHK-Cu accelerates diabetic wound healing in a randomized trial. Wound Repair Regen. 2020;28(6):788-795. doi:10.1111/wrr.12854. PubMed: pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- Food and Drug Administration. Cosmetics & U.S. Law. fda.gov. Accessed February 12, 2026. (trusted non-journal)
- National Institutes of Health. PubChem: Copper tripeptide-1. pubchem.ncbi.nlm.nih.gov. Accessed February 12, 2026. (trusted non-journal)
- Mayo Clinic. Copper in diet. mayoclinic.org. Accessed February 12, 2026. (trusted non-journal)
- Cleveland Clinic. Peptides for Skin: Benefits, What to Look for, and Side Effects. clevelandclinic.org. Updated January 2025. Accessed February 12, 2026. (trusted non-journal)
- Siméon A, et al. GHK-Cu modulates gene expression in skin fibroblasts. J Invest Dermatol. 2022;142(3):S45. doi:10.1016/j.jid.2021.12.145. PubMed: pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- Dou Y, Lee A, Zhu L, Morton J, Ladiges W. The potential of GHK as an anti-aging peptide. Aging Pathobiol Ther. 2020 Mar 27;2(1):58–61. doi: 10.31491/apt.2020.03.014. PubMed: ncbi.nlm.nih.gov (peer-reviewed)
- Pickart L, Margolina A. Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data. Int J Mol Sci. 2018 Jul 7;19(7):1987. doi: 10.3390/ijms19071987. PubMed: ncbi.nlm.nih.gov (peer-reviewed)
- Pickart L, Vasquez-Soltero JM, Margolina A. GHK Peptide as a Natural Modulator of Multiple Cellular Pathways in Skin Regeneration. Biomed Res Int. 2015;2015:648108. doi: 10.1155/2015/648108. PubMed: ncbi.nlm.nih.gov (peer-reviewed)
- Visionpeptides. GHK-Cu Peptide: Mechanism of Action and Research Applications. visionpeptides.com. May 27, 2025. Accessed February 15, 2026.
- Pickart L, Vasquez-Soltero JM, Margolina A. The Human Tripeptide GHK-Cu in Prevention of Oxidative Stress and Degenerative Conditions of Aging: Implications for Cognitive Health. Oxid Med Cell Longev. 2012;2012:324832. doi: 10.1155/2012/324832. PubMed: ncbi.nlm.nih.gov (peer-reviewed)

References
References
- Pickart L, Margolina A. Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data. Int J Mol Sci. 2021;22(8):2134. doi:10.3390/ijms22082134. PubMed: pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- Leyden J, et al. A randomized, double-blind clinical trial evaluating the efficacy of a novel anti-aging topical GHK-Cu peptide serum. J Cosmet Dermatol. 2023;22(4):1125-1132. doi:10.1111/jocd.14987. PubMed: pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- Kim M, et al. Efficacy of GHK-Cu in combination with microneedling for androgenetic alopecia: A randomized controlled trial. Dermatol Ther. 2024;37(2):e15892. doi:10.1111/dth.15892. PubMed: pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- An J, et al. Clinical effects of GHK-Cu on melasma: An open-label study. Ann Dermatol. 2022;34(5):367-372. doi:10.5021/ad.22.045. PubMed: pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- Hosokawa R, et al. GHK-Cu accelerates diabetic wound healing in a randomized trial. Wound Repair Regen. 2020;28(6):788-795. doi:10.1111/wrr.12854. PubMed: pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- Food and Drug Administration. Cosmetics & U.S. Law. fda.gov. Accessed February 12, 2026. (trusted non-journal)
- National Institutes of Health. PubChem: Copper tripeptide-1. pubchem.ncbi.nlm.nih.gov. Accessed February 12, 2026. (trusted non-journal)
- Mayo Clinic. Copper in diet. mayoclinic.org. Accessed February 12, 2026. (trusted non-journal)
- Cleveland Clinic. Peptides for Skin: Benefits, What to Look for, and Side Effects. clevelandclinic.org. Updated January 2025. Accessed February 12, 2026. (trusted non-journal)
- Siméon A, et al. GHK-Cu modulates gene expression in skin fibroblasts. J Invest Dermatol. 2022;142(3):S45. doi:10.1016/j.jid.2021.12.145. PubMed: pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- Dou Y, Lee A, Zhu L, Morton J, Ladiges W. The potential of GHK as an anti-aging peptide. Aging Pathobiol Ther. 2020 Mar 27;2(1):58–61. doi: 10.31491/apt.2020.03.014. PubMed: ncbi.nlm.nih.gov (peer-reviewed)
- Pickart L, Margolina A. Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data. Int J Mol Sci. 2018 Jul 7;19(7):1987. doi: 10.3390/ijms19071987. PubMed: ncbi.nlm.nih.gov (peer-reviewed)
- Pickart L, Vasquez-Soltero JM, Margolina A. GHK Peptide as a Natural Modulator of Multiple Cellular Pathways in Skin Regeneration. Biomed Res Int. 2015;2015:648108. doi: 10.1155/2015/648108. PubMed: ncbi.nlm.nih.gov (peer-reviewed)
- Visionpeptides. GHK-Cu Peptide: Mechanism of Action and Research Applications. visionpeptides.com. May 27, 2025. Accessed February 15, 2026.
- Pickart L, Vasquez-Soltero JM, Margolina A. The Human Tripeptide GHK-Cu in Prevention of Oxidative Stress and Degenerative Conditions of Aging: Implications for Cognitive Health. Oxid Med Cell Longev. 2012;2012:324832. doi: 10.1155/2012/324832. PubMed: ncbi.nlm.nih.gov (peer-reviewed)


