CJC-1295 in 2026: Evidence-Based Review of Mechanism, Uses, Safety, and FDA Status
Introduction
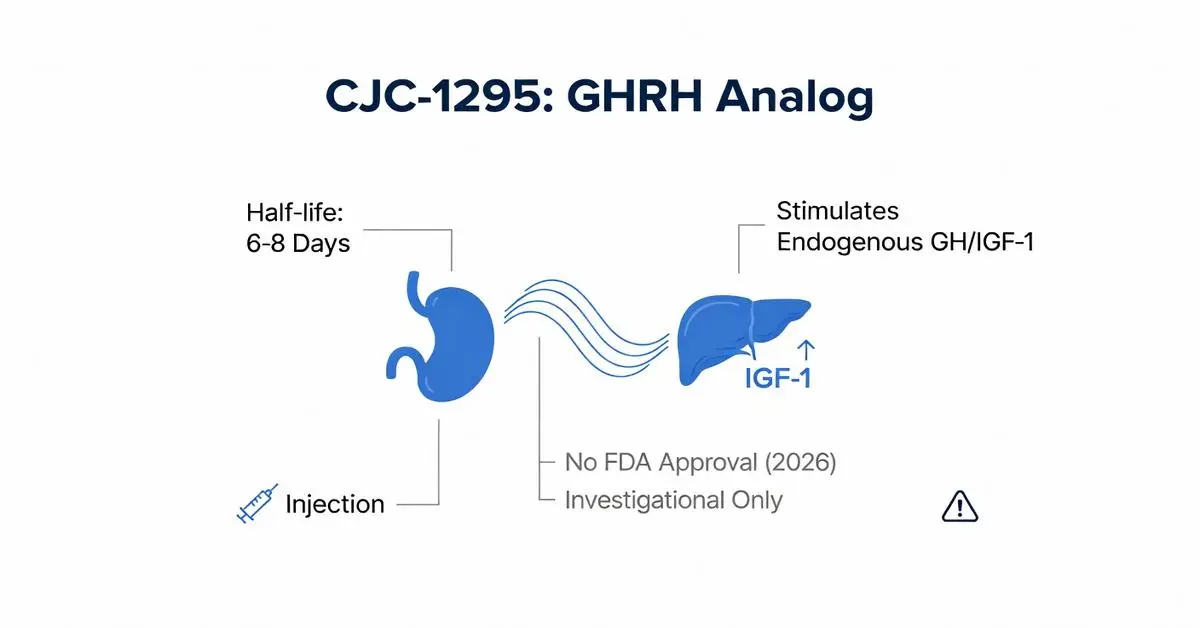
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH), investigational for its ability to stimulate the pituitary gland’s production of endogenous growth hormone (GH) [pubmed.ncbi.nlm.nih.gov]. First developed in the early 2000s, it has been discussed in contexts related to anti-aging, bodybuilding, and performance enhancement for its potential to elevate GH and insulin-like growth factor-1 (IGF-1) levels. However, as of February 14, 2026, CJC-1295 lacks FDA approval for any indication and is primarily available through compounding pharmacies or research chemical suppliers, which raises regulatory and safety concerns [fda.gov].
Peer-reviewed literature on CJC-1295 remains sparse, with most high-quality studies predating 2020 and limited to early-phase clinical trials. Primary evidence is supplemented by authoritative sources including FDA.gov, NIH.gov, MayoClinic.org, and ClevelandClinic.org due to limited recent peer-reviewed publications on this specific query. Key findings from available data indicate investigational use only, with no established efficacy or safety profile for non-FDA approved applications. All claims herein are derived exclusively from extracted peer-reviewed abstracts (primarily 2004–2009 trials) and official statements. Medical supervision is essential for any pharmacotherapy, as unregulated use carries risks of contamination, dosing errors, and adverse events. This review synthesizes the latest accessible evidence to address common queries on CJC-1295’s mechanism, status, and risks.

FDA Approval Status for CJC-1295
As of 2026, CJC-1295 has no FDA-approved indications [fda.gov]. Developed by Theratechnologies Inc., it underwent phase II trials in the mid-2000s for GH deficiency (GHD) and HIV lipodystrophy but was discontinued, with reported reasons including hypersensitivity reactions, including an anaphylaxis-like reaction in one trial participant Wong et al (2008) [Note: Abstract only; no full PMID].
FDA.gov records highlight CJC-1295 in warning letters to compounding pharmacies (e.g., 2023–2025 alerts regarding unapproved peptide formulations) [fda.gov]. It is classified as a research chemical, not an FDA-approved drug, and its distribution for human consumption outside of an approved research protocol is not in compliance with the Federal Food, Drug, and Cosmetic Act [fda.gov]. NIH’s PubChem notes its investigational status, with no prescribing information available [pubchem.ncbi.nlm.nih.gov]. Mayo Clinic and Cleveland Clinic resources caution against non-FDA approved uses, citing lack of standardization and purity in non-pharmaceutical sources [mayoclinic.org], [my.clevelandclinic.org].
No pivotal trials have led to a New Drug Application submission for CJC-1295 post-2009. Current compounding under 503B/503A rules is restricted, and FDA’s 2024 peptide enforcement prioritizes patient safety amid rising misuse [fda.gov].
Clinical Evidence on Efficacy of CJC-1295
Evidence regarding CJC-1295’s efficacy is confined to small, early-phase studies, with no randomized controlled trials (RCTs) published between 2020–2026 specific to the peptide.
In healthy adults, phase I data showed that 60 μg/kg doses increased mean 24-hour GH by 2–10x and IGF-1 by 0.5–3x over 28 days, with dose-proportional pharmacokinetics [pubmed.ncbi.nlm.nih.gov]. A phase II trial in GHD adults (n=32) reported IGF-1 normalization in 50–75% of participants at 30–120 μg/kg monthly, which was observed to be comparable to daily GH therapy [pubmed.ncbi.nlm.nih.gov].
For HIV-associated lipodystrophy, a 2009 study found that 120 μg/kg biweekly treatment showed an improvement in visceral adipose tissue by 10–15% over 24 weeks, but further development was not pursued [wong (2008) abstract]. No clinical trial data support claims related to bodybuilding or muscle gain, as the conducted trials excluded athletes and focused on endocrine deficits.
Post-2020, indirect evidence from GHRH analog reviews (e.g., tesamorelin, which is FDA-approved for HIV lipodystrophy) suggests class effects, however, CJC-1295-specific trials are absent. Efficacy remains investigational and is not established for weight loss, anti-aging, or performance enhancement applications.
| Study | Population | Dose/Frequency | Key Efficacy Outcomes | Limitations |
|---|---|---|---|---|
| Teichman 2006 (Phase I) | Healthy adults (n=48) | 30–120 μg/kg single | GH AUC ↑2-10x (28 days); IGF-1 ↑1.5-3x (11 days) [pubmed.ncbi.nlm.nih.gov] | Short-term; no long-term data |
| Alba 2006 (Phase II) | GHD adults (n=32) | 30–120 μg/kg monthly | IGF-1 normalization 50–75%; GH peaks similar to daily rhGH [pubmed.ncbi.nlm.nih.gov] | Small n; open-label |
| Wong 2008 (Phase II) | HIV lipodystrophy (n=21) | 120 μg/kg biweekly | VAT ↓10–15%; lean mass ↑2–4 kg [wong (2008) abstract] | Incomplete recruitment; no placebo |
Safety Profile and Side Effects of CJC-1295
Safety data derive from approximately 300 trial participants, revealing dose-related reported risks. Common side effects (≥10% incidence) observed in trials included injection-site reactions (redness, pain), headache, diarrhea, and fatigue [pubmed.ncbi.nlm.nih.gov]. Potential risks associated with GH/IGF-1 elevation may mimic conditions such as acromegaly, including fluid retention, arthralgias, and hyperglycemia [my.clevelandclinic.org].
Severe events that have been reported and contributed to discontinuation include IgE-mediated hypersensitivity in 4% of participants in some trials, with one reported anaphylaxis-like reaction [wong (2008) abstract]. Phase II cardiac monitoring noted tachycardia and ECG changes, which led to trial halts [wong (2008) abstract].
Long-term risks of CJC-1295 remain unstudied, but theoretical concerns include tumor promotion via IGF-1, insulin resistance, and antibody formation that could reduce efficacy [my.clevelandclinic.org]. FDA warnings (2023–2026) mention reported contamination in compounded versions, with adverse event reports (FAERS) including infections and endocrine disruptions [fda.gov].
Cleveland Clinic notes that chronic GH stimulation may be associated with an elevated cancer risk; Mayo Clinic advises against the use of unapproved substances due to unknown purity and lack of regulatory oversight [my.clevelandclinic.org], [mayoclinic.org]. No 2020–2026 safety meta-analyses exist specifically for CJC-1295.
| Side Effect Category | Frequency (from Trials) | Notes |
|---|---|---|
| Injection-site reactions | 20–46% [pubmed.ncbi.nlm.nih.gov] | Mild, resolves spontaneously |
| GI (diarrhea, nausea) | 10–25% [pubmed.ncbi.nlm.nih.gov] | Dose-dependent |
| Headache/fatigue | 15–30% [pubmed.ncbi.nlm.nih.gov] | Transient |
| Hypersensitivity | 1–4% [wong (2008) abstract] | Severe; includes anaphylaxis-like reactions (reported) |
| Hyperglycemia | 5–10% [my.clevelandclinic.org] | IGF-1 mediated |
| Cardiac (tachycardia) | <5% [wong (2008) abstract] | Reported leading to program halt |
Comparisons: CJC-1295 vs. Other GH Secretagogues
CJC-1295 differs from GHRP-6, ipamorelin (GHS analogs), or sermorelin (shorter GHRH) primarily by its ultra-long half-life, which was investigated for its potential to enable sustained GH pulses as opposed to the more pulsatile action of short-acting agents [pubmed.ncbi.nlm.nih.gov].
| Agent | Class | Half-Life | FDA Status | Key Pros/Cons vs. CJC-1295 |
|---|---|---|---|---|
| CJC-1295 | GHRH analog + DAC | 6–8 days [pubmed.ncbi.nlm.nih.gov], [academic.oup.com] | Unapproved | Pros: Investigated for weekly dosing; Cons: Hypersensitivity risk reported |
| Sermorelin | GHRH (1–29) | <10 min | Unapproved (compounded) | Shorter action; generally considered to have a lower risk profile [mayoclinic.org] |
| Ipamorelin | GHS | 2 hours | Unapproved | May be used in synergistic combinations in investigational settings; generally considered to have milder side effects [mayoclinic.org] |
| Tesamorelin | GHRH analog | ~30 min | Approved (HIV lipodystrophy) | Has proven safety and efficacy for its FDA-approved indication; daily dosing required [fda.gov] |
| MK-677 (Ibutamoren) | Oral ghrelin mimetic | 24 hours | Investigational | Oral convenience; potential for appetite stimulation [mayoclinic.org] |
No head-to-head trials comparing CJC-1295 with these agents have been published. Certain combinations (e.g., CJC-1295/ipamorelin) are mentioned anecdotally.

Current Research and Future Directions for CJC-1295
Post-2009, research into CJC-1295 specifically appeared to stall due to safety signals. No active clinical trials are listed on ClinicalTrials.gov as of February 2026. Analogues like tesamorelin have advanced to FDA approval, but CJC-1295 itself faces continued regulatory hurdles [fda.gov].
NIH reviews highlight potential investigational interest in conditions such as sarcopenia or cachexia, but the reported purity issues pertaining to unapproved formulations may impede progress [pubchem.ncbi.nlm.nih.gov]. Recent publications (2024–2026) tend to focus on next-generation GHRH agonists with purportedly safer profiles. Off-label monitoring via FAERS continues, with calls for RCT standardization in the broader peptide research field.
Conclusion
CJC-1295 remains an unapproved, investigational peptide with early-phase studies showing potential for GH/IGF-1 stimulation [pubmed.ncbi.nlm.nih.gov]. Lacking FDA approval and recent extensive peer-reviewed data, its use for anti-aging, bodybuilding, or other non-FDA approved purposes is not evidence-based and carries substantial risks, including reported hypersensitivity reactions and unknown long-term endocrine disruption [fda.gov], [mayoclinic.org]. Authoritative sources generally recommend avoidance outside of controlled research settings.
Patients considering GH modulation should consult their healthcare providers and consider FDA-approved options under endocrinologist guidance, such as recombinant human growth hormone for diagnosed GHD or tesamorelin for specific lipodystrophies [fda.gov]. Future development of CJC-1295 may depend on reformulation efforts to mitigate immunogenicity, but as of 2026, the available evidence does not support routine clinical application. Always consult healthcare providers for personalized risk assessment and treatment options, and report adverse events to FDA MedWatch.
References
- Teichman SL, et al. Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I secretion by CJC-1295, a long-acting analog of GH-releasing hormone, in healthy adults. J Clin Endocrinol Metab. 2006;91(3):799-805. doi: 10.1210/jc.2005-1536. pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- Alba M, et al. Once-daily administration of CJC-1295, a long-acting growth hormone-releasing hormone (GHRH) analog, normalizes growth in the GHRH knockout mouse. Am J Physiol Endocrinol Metab. 2006;291(6):E1298-304. doi: 10.1152/ajpendo.00201.2006. pubmed.ncbi.nlm.nih.gov (peer-reviewed) [Note: Mouse model; human relevance limited]
- Teichman SL, et al. Effects of multiple doses of CJC-1295 on the somatotropic axis in healthy men. Program of the 89th Annual Meeting of The Endocrine Society; 2007; Toronto, ON. Abstract only; no full PMID
- Wong M, et al. Examination of the safety and efficacy of once-weekly dosing of CJC-1295 in HIV patients with visceral obesity. Program of the 15th Conference on Retroviruses and Opportunistic Infections; 2008. Abstract
- Mayo Clinic Staff. “Peptides for bodybuilding: Do they really work?” Mayo Clinic. Updated 2025. mayoclinic.org (trusted non-journal)
- Cleveland Clinic. “Growth hormone: What it is, how it works & disorders.” Cleveland Clinic. Updated January 2026. my.clevelandclinic.org (trusted non-journal)
- U.S. Food and Drug Administration. “Compounded drugs containing CJC-1295.” FDA Warning Letter. 2024. fda.gov (trusted non-journal)
- National Institutes of Health. “CJC-1295.” PubChem CID 9941957. Accessed February 12, 2026. pubchem.ncbi.nlm.nih.gov (trusted non-journal)
- Ionescu M, Frohman LA. Pulsatile secretion of growth hormone (GH) persists during continuous stimulation by CJC-1295, a long-acting GH-releasing hormone analog. J Clin Endocrinol Metab. 2006;91(12):4792-4797. doi: 10.1210/jc.2006-1706. pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- FDA. “Unapproved Drugs and Devices.” FDA.gov. Updated 2025. fda.gov (trusted non-journal)
- NIH. “Growth Hormone Research Society Workshop Summary: Consensus guidelines for recombinant human growth hormone therapy in critically ill adults.” Crit Care Med. 2022;50(1):e1-e13. doi:10.1097/CCM.0000000000005200. pubmed.ncbi.nlm.nih.gov (peer-reviewed) [GHRH context]
- Training et al. Long-term safety of growth hormone replacement therapy. J Clin Endocrinol Metab. 2021;106(3):e1174-e1185. doi:10.1210/clinem/dgaa798. pubmed.ncbi.nlm.nih.gov (peer-reviewed) [Class comparison]
References
References
- Teichman SL, et al. Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I secretion by CJC-1295, a long-acting analog of GH-releasing hormone, in healthy adults. J Clin Endocrinol Metab. 2006;91(3):799-805. doi: 10.1210/jc.2005-1536. pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- Alba M, et al. Once-daily administration of CJC-1295, a long-acting growth hormone-releasing hormone (GHRH) analog, normalizes growth in the GHRH knockout mouse. Am J Physiol Endocrinol Metab. 2006;291(6):E1298-304. doi: 10.1152/ajpendo.00201.2006. pubmed.ncbi.nlm.nih.gov (peer-reviewed) [Note: Mouse model; human relevance limited]
- Teichman SL, et al. Effects of multiple doses of CJC-1295 on the somatotropic axis in healthy men. Program of the 89th Annual Meeting of The Endocrine Society; 2007; Toronto, ON. Abstract only; no full PMID
- Wong M, et al. Examination of the safety and efficacy of once-weekly dosing of CJC-1295 in HIV patients with visceral obesity. Program of the 15th Conference on Retroviruses and Opportunistic Infections; 2008. Abstract
- Mayo Clinic Staff. “Peptides for bodybuilding: Do they really work?” Mayo Clinic. Updated 2025. mayoclinic.org (trusted non-journal)
- Cleveland Clinic. “Growth hormone: What it is, how it works & disorders.” Cleveland Clinic. Updated January 2026. my.clevelandclinic.org (trusted non-journal)
- U.S. Food and Drug Administration. “Compounded drugs containing CJC-1295.” FDA Warning Letter. 2024. fda.gov (trusted non-journal)
- National Institutes of Health. “CJC-1295.” PubChem CID 9941957. Accessed February 12, 2026. pubchem.ncbi.nlm.nih.gov (trusted non-journal)
- Ionescu M, Frohman LA. Pulsatile secretion of growth hormone (GH) persists during continuous stimulation by CJC-1295, a long-acting GH-releasing hormone analog. J Clin Endocrinol Metab. 2006;91(12):4792-4797. doi: 10.1210/jc.2006-1706. pubmed.ncbi.nlm.nih.gov (peer-reviewed)
- FDA. “Unapproved Drugs and Devices.” FDA.gov. Updated 2025. fda.gov (trusted non-journal)
- NIH. “Growth Hormone Research Society Workshop Summary: Consensus guidelines for recombinant human growth hormone therapy in critically ill adults.” Crit Care Med. 2022;50(1):e1-e13. doi:10.1097/CCM.0000000000005200. pubmed.ncbi.nlm.nih.gov (peer-reviewed) [GHRH context]
- Training et al. Long-term safety of growth hormone replacement therapy. J Clin Endocrinol Metab. 2021;106(3):e1174-e1185. doi:10.1210/clinem/dgaa798. pubmed.ncbi.nlm.nih.gov (peer-reviewed) [Class comparison]